Aging is not a singular process but a mosaic of biological interactions that unfold uniquely in each individual. Nonetheless, key principles may exist that allow us to map trajectories and uncover rejuvenating interventions. Current tools remain inadequate for early, personalized, and accurate risk assessment for neurodegeneration—either detecting risk after changes have already begun or failing to incorporate the interacting factors that drive disease risk. The key to improving the effectiveness of risk assessments is approaching what's happening in the brain at the molecular level. Without molecular readouts to detect early changes in brain health, clinicians and scientists are navigating in the dark until damage is already done and clinical symptoms appear.
Physician-scientist Fanny Elahi, MD, PhD, Associate Professor of Neurology, and Neuroscience, at the Icahn School of Medicine at Mount Sinai, and newly named Director of The Glickenhaus Center for Successful Aging (see related article here), is turning to the body’s universal messenger—the blood—to help illuminate this landscape. The Elahi Lab decodes the complex drivers of aging using blood-based biomarkers as molecular compasses to guide interventions. Ultimately, by focusing on the blood and vasculature, these molecular signatures could allow clinicians to chart personalized therapeutic interventions that restore homeostasis, support cognitive resilience, and extend the brain’s healthspan.
Using artificial intelligence and machine learning, the lab extracts molecular signatures from large-scale proteomic datasets. These data-driven profiles—derived primarily from blood and linked to cerebrospinal fluid and brain tissue—hold promise as accessible, scalable biomarkers that reflect the brain’s underlying state.
Recent work from the lab marks a major step toward this goal, identifying vascular proteomic signatures in blood and cerebrospinal fluid that predict adverse brain aging trajectories and vulnerability to neurodegeneration. Published and preprinted work from the lab (Torres-Espin et al., 2024; Keller et al., 2024) indicate that vascular dysfunction may be an early and key driver of brain aging, and perhaps an ideal target for therapeutic interventions.
Importantly, the research also shows population-specific differences, including divergent brain trajectories for men and women. While the mechanisms underlying the differences are not yet fully understood, the Elahi Lab suspects that hormonal shifts in midlife may play a role in women’s increased vulnerability to Alzheimer’s disease—an area now under active study.
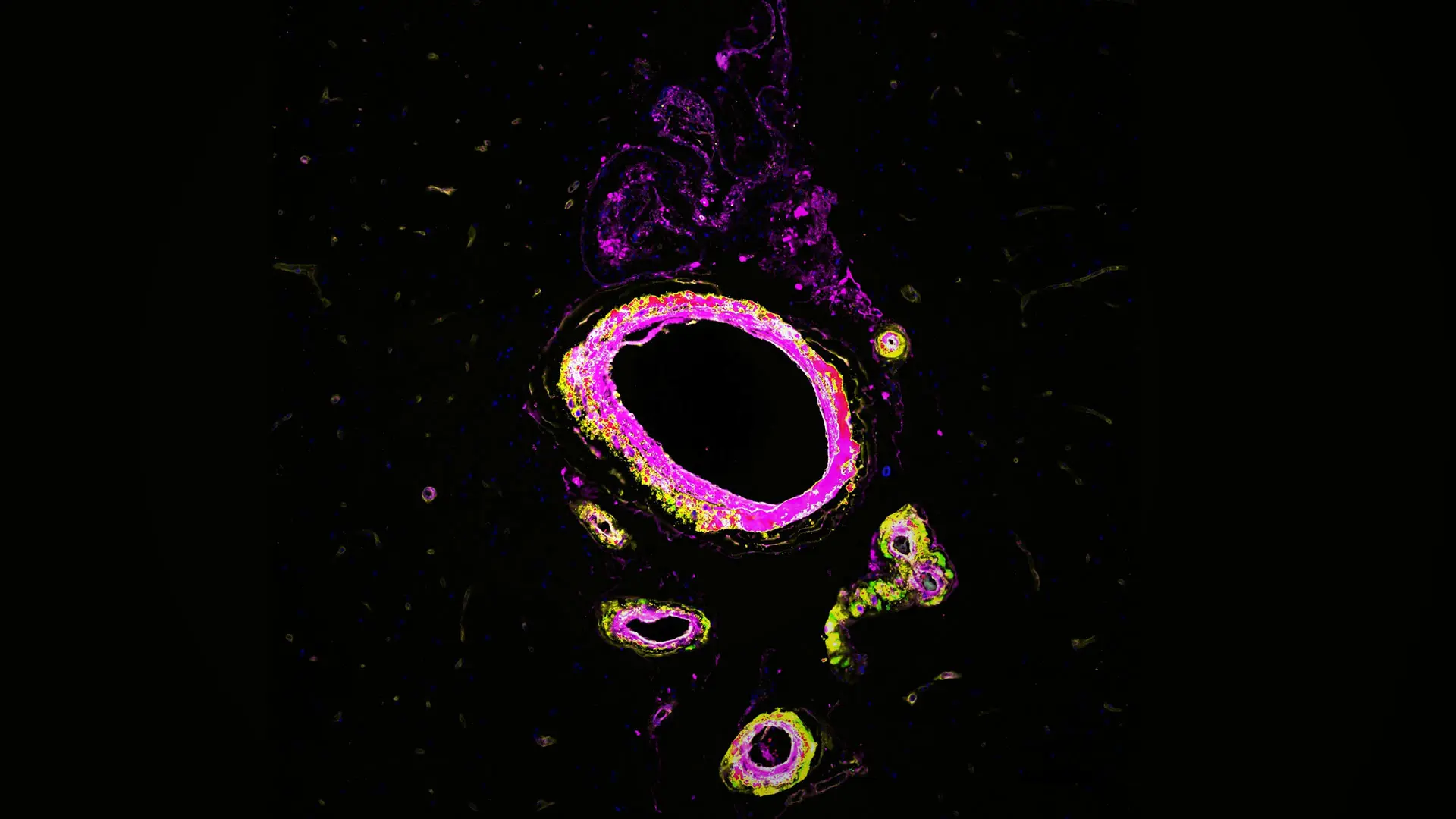
The lab’s unique approach does not stop at peripheral blood signatures of adverse brain aging. By examining postmortem human brain tissue in parallel with examining molecular biomarkers in blood, researchers in the Elahi Lab have shown that the same key proteins in blood signatures are expressed in the brain’s vascular cells, and their levels are dysregulated in individuals with increased genetic risk for brain vascular aging, strokes, and cognitive decline (see Figure).
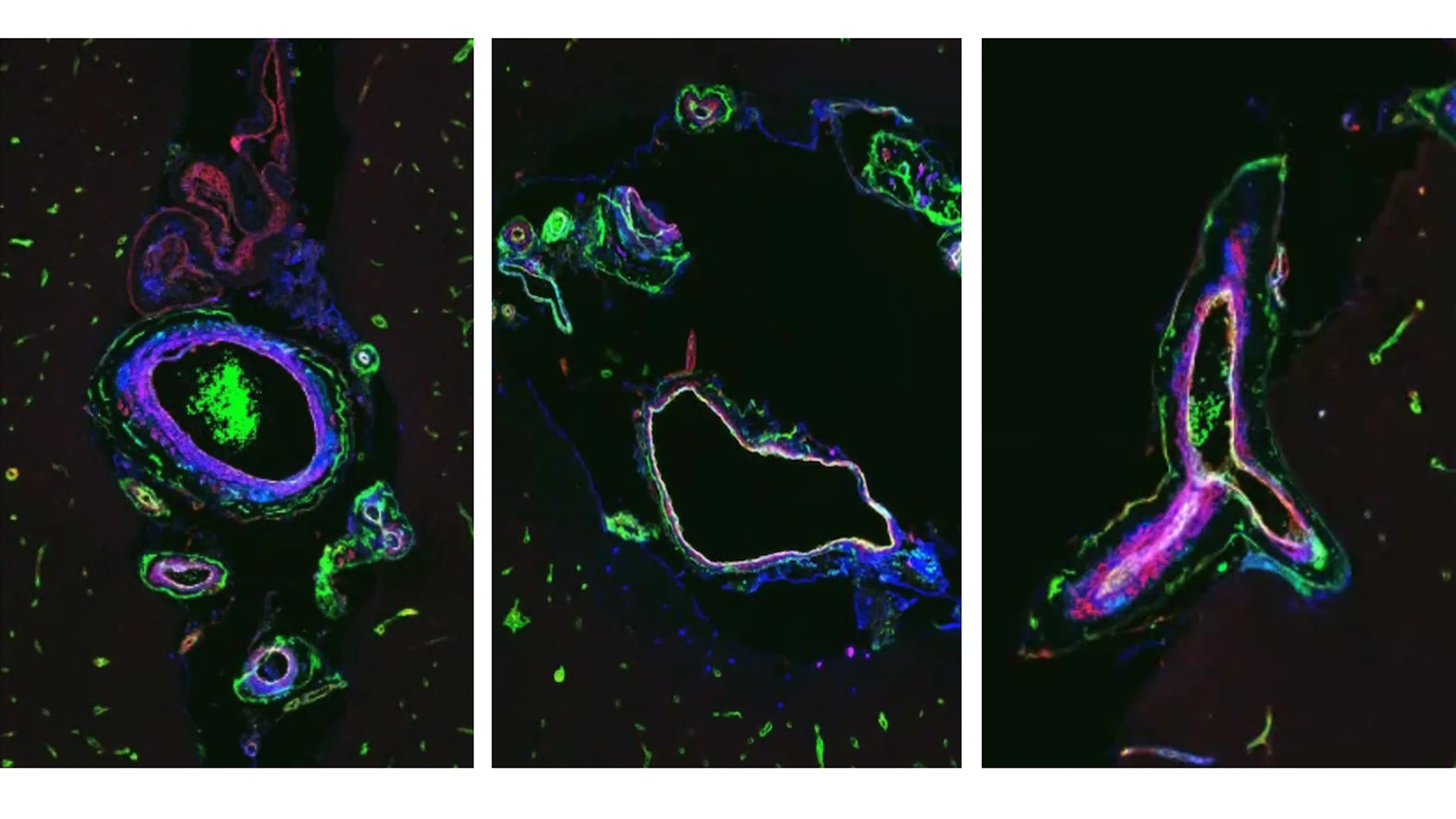
Figure. Multiplex immunohistochemistry reveals expression of key proteins from the vascular disease blood signature within brain blood vessels of a brain impacted by a monogenic form of accelerated vascular aging and dementia. DAPI marking nuclei (blue), C4A (green), FN1 (yellow), VEGFR1 (red), and CD105 (light blue).
Early blood signatures that predict brain aging trajectories combined with strong expression of these proteins in the brain blood vessels of individuals with elevated risk for dementia suggests that vascular health plays a pivotal role in overall brain health. To that end, the Elahi Lab is building a vascular cell atlas of vulnerable human brains—mapping how aging and vascular genetic risk alter the function of specific endothelial and mural cell populations. This foundational work links blood signatures to cellular identities and function. Anchoring these data to genetic risk enhances causal inference and moves research beyond correlative work and toward actionable steps to build resilience.
The Elahi Lab is building a vascular cell atlas of vulnerable human brains—mapping how aging and vascular genetic risk alter the function of specific endothelial and mural cell populations.
The group is also developing blood signatures to understand how upstream risk factors—including genetics, lifestyle, and systemic processes such as menopause, immune aging, hypertension, and metabolic dysregulation—modulate brain resilience or accelerate decline. The goal is to trace these pathways to their origins, identify molecular levers that could restore and maintain healthy brain function, and to create blood biomarker panels that can empower future clinical trials.
Now, a paradigm shift is underway—one that bridges biology, diagnostics, and therapeutics to protect the aging brain. Achieving this vision requires close integration between biomarker science and therapeutic development. The Elahi Lab is helping to lead this transformation, recognizing that the true power of blood-based biomarkers lies not in what they reveal in isolation but in how they integrate into a broader framework that enables early detection and precision intervention.
Featured

Fanny Elahi, MD, PhD
Associate Professor of Neurology, and Neuroscience, Icahn School of Medicine at Mount Sinai, and Director, The Glickenhaus Center for Successful Aging