Clinical trials urgently need a paradigm shift in their designs if physicians and scientists are to understand better how cancer immunotherapies work and improve patient outcomes across tumor types. That premise is the backbone of an array of TCI multidisciplinary programs, including the Early Phase Trial Unit (EPTU) and The neoAdjuvant Research Group to Evaluate Therapeutics (TARGET), which bridge expertise with researchers from the Marc and Jennifer Lipschultz Precision Immunology Institute (PrIISM) at the Icahn School of Medicine at Mount Sinai.
EPTU emphasizes first-in-human and biomarker-focused proof-of-concept studies, actively promoting the translation of scientific discoveries from TCI laboratories to clinical applications. New agents may be piloted or used in novel combinations with other agents, or they may be administered in ways hypothesized to be more effective, such as injection directly into tumors.
TARGET, too, focuses on first-in-human studies, but occupies the neoadjuvant space. It generates multidimensional datasets that can inform future development of immunotherapies by harnessing Mount Sinai’s considerable expertise and resources in the fields of surgery, pathology, radiology, genomics, immunology, and data science.

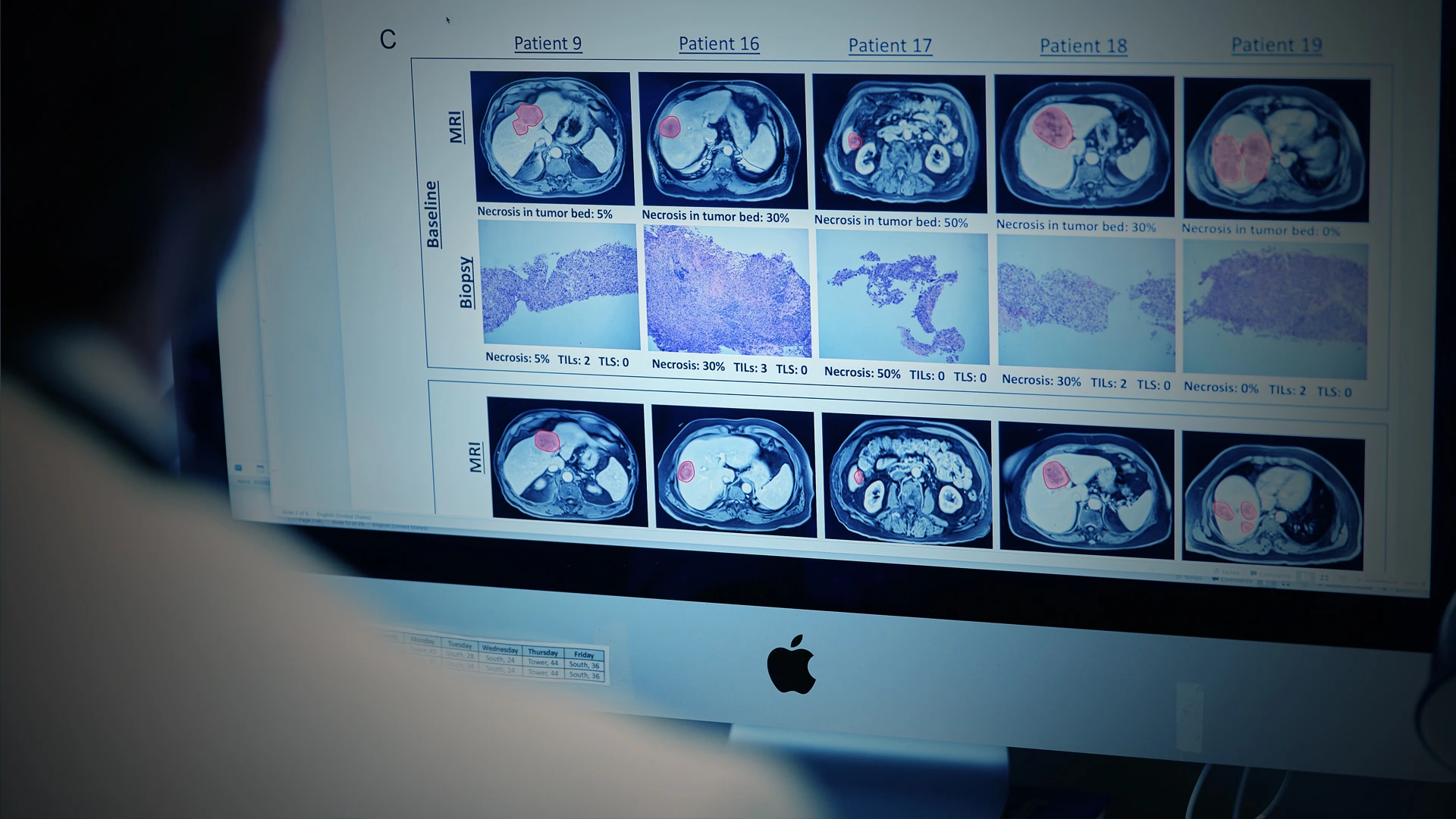
The EPTU and TARGET teams have been studying cancer therapies in “window-of-opportunity” trials, in which patients receive brief presurgical therapeutic interventions. The aim is to determine if these short-duration therapies help the immune system eliminate disease components that might spread before surgery, increasing the likelihood of a post-surgical cure. The trials also aim to study the effect of novel drugs on tumors and the immune system. Findings from one such trial were published in January 2022 in Lancet Gastroenterology and Hepatology.
“We have all these platforms that allow us to do a deep dive into the tissue and blood taken from patients in clinical trials, thus enabling us to characterize how these drugs work,” says Thomas Marron, MD, PhD, Director of EPTU and co-leader of TARGET.

The interdisciplinary collaboration between Thomas Marron, MD, PhD, and the Marc and Jennifer Lipschultz Precision Immunology Institute on TARGET aims to reduce the time it takes for therapeutics to reach patients.
The smaller and shorter-duration trials provide for extensive molecular and cellular characterizations to rapidly identify mechanisms of drug response and resistance. “This opens the door to identifying biomarkers that tell us who is going to respond to a drug and who is not, while informing our attempts to combine new therapies to improve responses, ideally without compounding toxicity,” Dr. Marron says.
Pushing Clinical and Equitable Success
A TARGET/EPTU study, characterized in Lancet Gastroenterology and Hepatology, evaluated clinical activity of neoadjuvant anti-PD1 monotherapy in patients with resectable hepatocellular carcinoma.
The phase 2 trial had 21 patients who received two cycles of cemiplimab (350 mg) intravenously every three weeks, followed by resection. After surgery, participants received an additional eight cycles of the PD1 immunotherapy every three weeks. The findings were encouraging.
“We were able to show significant clinical activity in a patient population that typically experiences a 70 percent chance of cancer recurrence following surgery, and for which no FDA-approved drug has demonstrated the ability to lower this poor prognosis,” reports Dr. Marron, lead author of the paper, and Associate Professor of Medicine (Hematology and Medical Oncology) at Icahn Mount Sinai.

Not only is Deborah Doroshow, MD, PhD, pushing the boundaries of oncological science, she is ensuring these innovations benefit underserved populations as well.
In addition to bringing cutting-edge science to the bedside, the study also took into account the social, physical, and emotional needs of trial participants. “We have developed a program that tends to the holistic needs of patients through integration with our colleagues in social work and supportive oncology,” says Deborah Doroshow, MD, PhD, Assistant Professor of Medicine (Hematology and Medical Oncology), who is a Principal Investigator in EPTU.
Dr. Doroshow has made it a priority for EPTU and TARGET trials to address inequities in underrepresented populations that have evolved over the years in cancer care. Patients from minority populations are typically vastly underrepresented in oncology clinical trials. However, nearly 50 percent of patients in EPTU’s early phase trials are people of color.
“In line with national guidelines,” Dr. Doroshow notes, “we firmly believe clinical trials should be offered to every patient at every juncture of their treatment because they might offer treatment options better than the standard of care they would otherwise receive.”
Featured

Deborah Doroshow, MD, PhD
Assistant Professor of Medicine (Hematology and Medical Oncology)

Thomas Marron, MD, PhD
Director of the Early Phase Trials Unit in The Tisch Cancer Institute