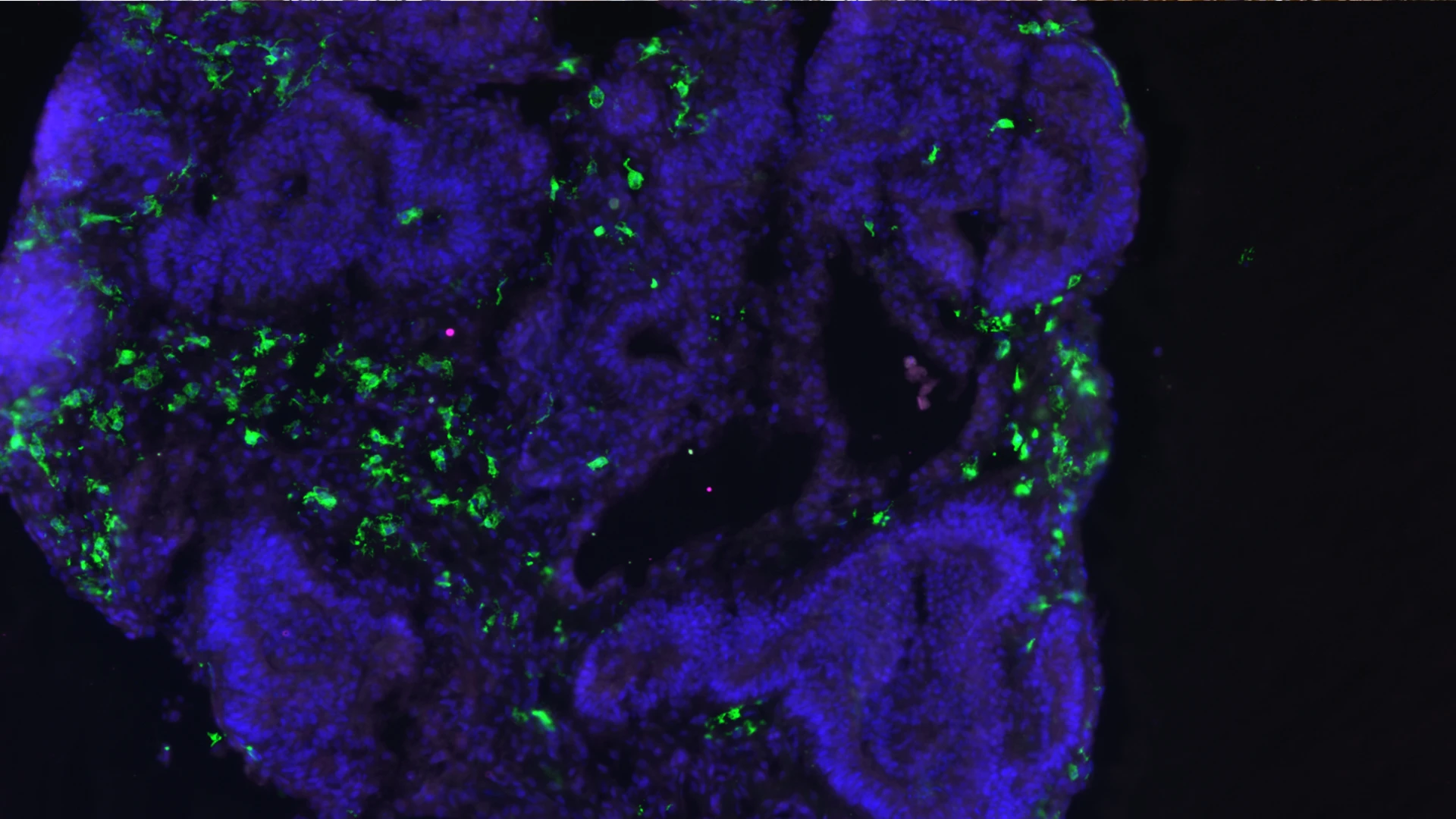
An organoid from Dr. De Witte’s lab, with the microglia shown in green. Image courtesy of Ormel PR et al., via Nature Communications.
Lotje de Witte, MD, PhD, Assistant Professor of Psychiatry at the Icahn School of Medicine at Mount Sinai, and her lab have become leading experts in the rapidly expanding field of cerebral organoids. Their work is adding to a body of knowledge about how the human immune system and its resident microglia contribute to neurodegenerative and neurodevelopmental disorders.
Dr. de Witte’s stem cell-derived organoid models are, for the first time, allowing scientists to study the cellular development and intercellular interactions among microglia and other cell types within a 3D human brain microenvironment. This has led to the discovery
that these cells can innately develop within cerebral organoid models, providing a vivid window with which to study the structural, functional, and molecular mechanisms underlying microglia development, function, and dysfunction in the human brain.
Scientists have known for some time that the immune system is involved in neurological and psychiatric conditions, and the mechanistic insights now offered by organoid models could potentially lead to screening for, and the development of, important new therapies for patients. Cerebral organoids are generated by culturing embryonic stem cells or
induced pluripotent stem cells into small balls of human brain-like tissue. With the help of a biochemical cocktail of proteins and minerals they grow and self-assemble in the lab dish, mimicking development of the fetal brain in the womb and forming specialized central nervous system regions such as cortex, hippocampus, and retina.
This video shows a cerebral organoid, with the dividing cells in red. Courtesy of Dilara Ilhan
A major thrust of Dr. de Witte’s research is exploring the impact of the environment on disease. “We know that disorders such as autism and schizophrenia are partially caused by genetic factors, but cerebral organoids are allowing us to study their interaction with
environmental triggers,” she says. In a study now underway, the team is exposing cerebral organoids to dietary changes in amino acid composition and has observed a reduction in cell size, as well as the upregulation of inflammatory pathways. Through such findings, the researchers hope to learn what cellular and molecular mechanisms are disrupted in neurodegenerative and neurodevelopmental diseases.
“I think we’re close to finding these mechanisms in Alzheimer’s disease and autism, and especially in the syndromal forms of neurodevelopmental disorders,” Dr. de Witte says. “And once we know which molecular pathways are affected we could be a step closer to screening for drugs that could mediate those pathways.”
The key to these breakthroughs will be the development of human organoid models that can help answer specific questions about the human brain. How are microglia involved in the development of human neurons and astrocytes, including processes such as neurogenesis and synaptic pruning? Can these models be optimized to help scientists better understand the role of microglia, macroglia, and neurons in neurodevelopmental diseases, particularly those such as autism and schizophrenia that have a clear genetic component?
“Cerebral organoids are very new and very exciting, and one of the biggest challenges facing the field is making sure our models are as robust as possible,” says Dr. de Witte. “Once we’ve mastered that we’ll be in a better position to know how neuron-microglia interactions impact the development, physiology, and pathology of the human brain.”
Featured

Lotje De Witte, MD, PhD
Assistant Professor of Psychiatry