With Janus kinase (JAK) inhibitors increasingly becoming an important tool for treating inflammatory diseases, PrIISM researchers have done extensive work in this field, including clinical studies in dermatitis and inflammatory bowel disease by physician-scientists Emma Guttman-Yassky, MD, PhD, and Jean-Frederic Colombel, MD.
The institute ramped up efforts in this area of research, and in January 2022, Mount Sinai recruited Brian Kim, MD, to serve as Professor of Dermatology and Vice Chair of Research at the Icahn School of Medicine at Mount Sinai. His work includes clinical trials of JAK inhibitors to treat skin conditions that cause chronic itch and also at the basic science level.
These kinase enzymes control inflammation, the biological process that moves immune cells from the blood into infected or damaged tissues to fight pathogens or mend wounds. But if JAKs are not switched off properly, inflammation can become chronic and cause disease in organs and tissues throughout the body.
Dr. Kim and his team had been studying communication between the immune system and neurons in parts of the body outside the brain and how it affects inflammation and sensation. They then found that JAK activity controls itch in an unexpected way: both chronic and acute itch are caused by the skin’s sensory neurons’ response to inflammatory proteins, rather than immune cells in the skin. Such JAK activity in neurons was discovered to be key to the constant chronic itching in diseases such as atopic dermatitis.
Earlier this year, the U.S. Food and Drug Administration approved the use of JAK inhibitors as treatment for symptoms of autoimmune diseases such as atopic dermatitis, alopecia areata, and vitiligo. The use of such drugs to treat itch has transformed patients’ lives.
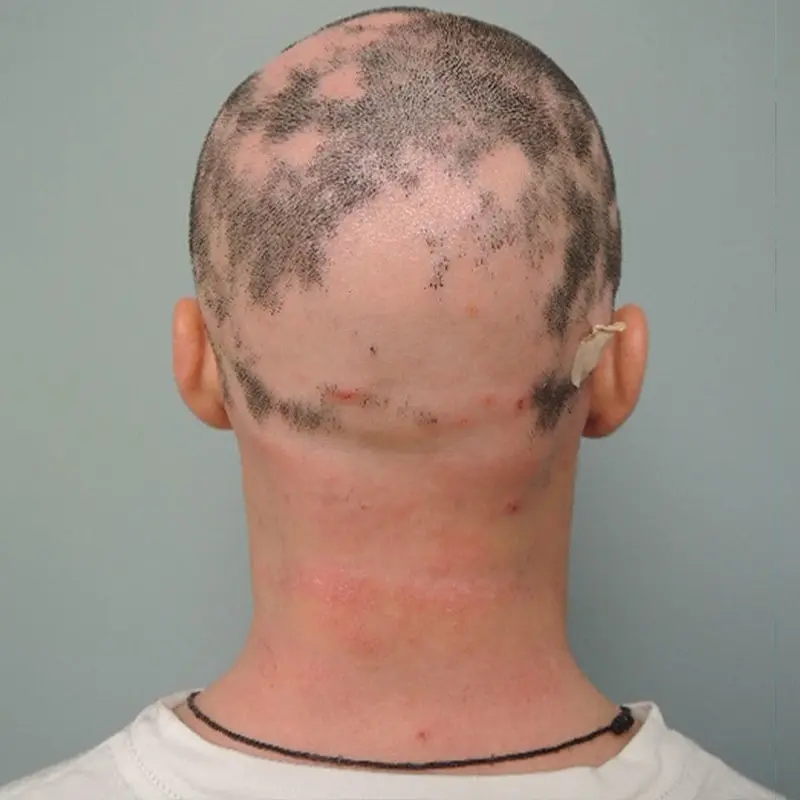
Patient with alopecia areata before treatment with JAK inhibitor
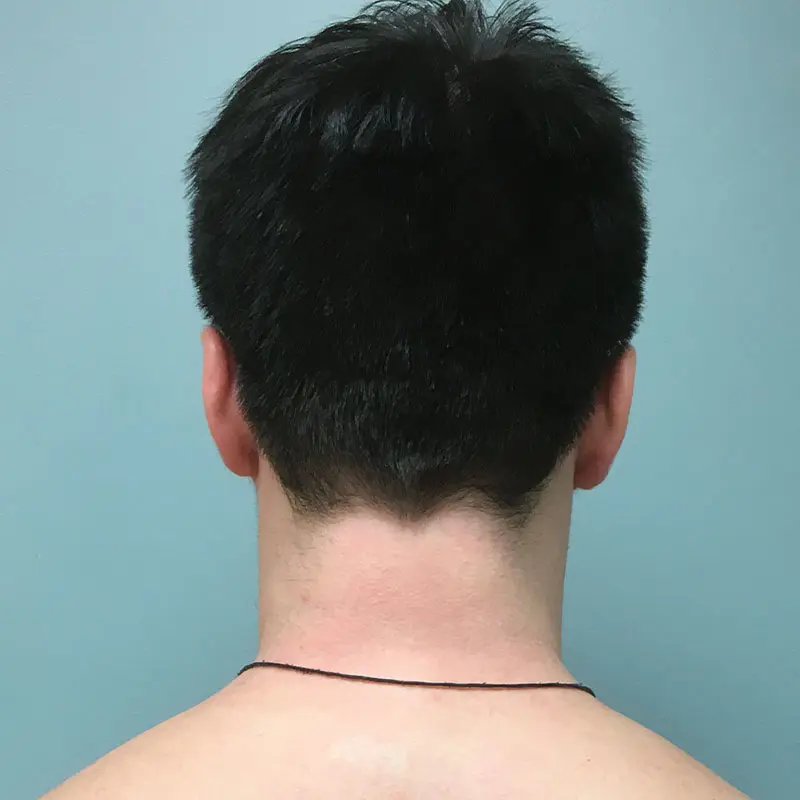
Patient with alopecia areata after treatment with JAK inhibitor
Dr. Kim’s work has found global affirmation—he recalled an email from Stuart Turvey, MBBS, DPhil, FRCPC, a professor of pediatrics at the University of British Columbia, who identified the first germ line gain-of-function JAK1 mutation in humans, which said “I think your discovery is real.” Dr. Turvey told Dr. Kim that his pediatric patients who had a JAK mutation never stopped itching their whole lives and couldn’t even sleep very well. But when given a JAK1/2 inhibitor, the children finally slept through the night. “Before that, nothing helped,” Dr. Turvey had told Dr. Kim.
Building on Turvey’s insight, Dr. Kim then took the mutation identified in Dr. Turvey’s patients and inserted it into a mouse to recapitulate the phenotype. “We are now using that mouse to figure out how JAK1 activity in the nerves in the skin causes itch,” Dr. Kim said.

Dusan Bogunovic, PhD, and Brian Kim, MD
Complex Roles for JAKs in Disease
Although JAK activity is critical for inflammation in many diseases, it is unclear why that activity is not dialed down appropriately in the first place. New clues to the source of JAK dysfunction in disease are coming from the lab of PrIISM researcher Dusan Bogunovic, PhD, Director of the Center for Inborn Errors of Immunity.
A few years ago, Jeffrey Saland, MD, Chief of the Division of Pediatric Nephrology and Hypertension, referred a complex patient to the Undiagnosed Disease Patient Program, of which Dr. Bogunovic is a part. Dr. Bogunovic’s team identified a causative JAK1 mutation in this patient and did an extensive molecular, biochemical, and immunological work-up. The findings led the patient’s gastrointestinal specialist to treat with a JAK1 inhibitor, which transformed the patient’s life. “While this mutation in JAK1 was very impactful, we thought that maybe other mutations in JAK1 exist, and that they could cause a spectrum of disease manifestations,” Dr. Bogunovic said.
Since then, Dr. Bogunovic’s team has discovered a variety of unknown variants in the JAK1 gene by sequencing patient samples from the BioMe BioBank at Icahn Mount Sinai. Preliminary analyses suggest several variants correlate with increased risk of specific pathologies.
“We identified hundreds of patients with putative mutations in JAK1,” said Dr. Bogunovic, adding that his team has to characterize each mutation molecularly, biochemically, and immunologically to determine its pathogenic potential. “Having said that, these mutations are still exceedingly rare. None of the genetic variations identified is more frequent than about 1 in 2,000 individuals. Collectively, however, they could explain up to half of a percent of the patients’ conditions.”
Not all JAK1 mutations are made equal, and Dr. Bogunovic’s lab is actively examining how the hundreds of JAK1 variants lead to different disease symptoms and pathologies. Furthermore, with Dr. Kim joining PrIISM and his work complementing Dr. Bogunovic’s, both researchers are working together to unravel how JAKs operate in different cell types and tissues to determine disease.

Drs. Bogunovic and Kim with lab member Ichiro Imanishi discussing molecule design
The researchers are examining not just where mutations are found within the JAK protein, but also the specific amino acids that are changed that determine how the variants influence kinase activity. The teams are also looking at where and when JAK1 is expressed, especially among different tissues and cell types.
“One variant may lead to the augmentation of canonical pathways for inflammation, but others may lead to the introduction of novel signaling cascades,” Dr. Bogunovic said. “This information will be incredibly important when choosing the appropriate JAK inhibitor.”
Featured

Dusan Bogunovic, PhD
Professor, Oncological Sciences, Microbiology, Dermatology, Pediatrics

Brian S. Kim, MD
Director of the Mark Lebwohl Center for Neuroinflammation and Sensation
