The Blavatnik Family Women’s Health Research Institute, led by Director Leslee Shaw, PhD, draws its team of faculty from many departments at the Icahn School of Medicine at Mount Sinai. The teams perform state-of-the-art research across the woman’s life span, with the goal of filling in knowledge gaps and informing advanced and compassionate care.
In addition, the Center of Excellence for Gynecologic Cancer, led by Co-Directors Stephanie V. Blank, MD, and Dmitriy Zamarin, MD, PhD, provides access to novel therapies through clinical trials, along with a comprehensive range of services that includes cancer screening and diagnosis, treatments, and supportive services.
Below, members of the research teams provide insights into selected 2025 publications:
“Characterizing Labor Progression and Duration According to Maternal Body Mass Index,” American Journal of Obstetrics & Gynecology, November 2025.
Sara Edwards, MD, Rebecca Cohen, Zhan Zhao, MA, Raina Kishan, MD, Morgan Steelman, Angela Bianco, MD, Katharine McCarthy, PhD, MPH, Kimberly B. Glazer, PhD, MPH, Chelsea A. DeBolt, MD.
Abstract: The objective was to describe labor progression and duration according to maternal body mass index in patients who achieve vaginal delivery.
This was a retrospective cohort study of all vaginal deliveries at a large, tertiary hospital in New York City from January 1, 2013, through December 31, 2022. We included singleton births of cephalic-presenting fetuses delivering vaginally at gestational age ≥ 37 weeks, with at least 2 documented cervical exams between 2 and 10 cm dilation. Body mass index was documented from delivery admission or within 30 days prior and classified according to Institute of Medicine categories. All demographic information, clinical characteristics, cervical exams, and obstetric outcomes were abstracted from the electronic medical record. Outcomes included length of latent phase, active phase, and total first stage of labor. Labor curves were constructed for each body mass index group and further subcategorized by parity, as well as induction versus spontaneous labor.
Among 41,868 deliveries that met inclusion criteria, 9,491 were normal weight or underweight (22.7 percent), 19,658 were overweight (47 percent percent), 8,880 had class 1 obesity (21.2 percent), 2,826 class 2 obesity (6.7 percent), and 1,013 class 3 obesity (2.4 percent). Labor duration differed by body mass index class, with a positive monotonic pattern observed between increasing body mass index class and time in latent labor and first stage of labor. Duration of active labor did not differ among individuals with overweight or obesity compared to normal weight. Results were similar in sub-group analyses based on parity and presence of spontaneous labor versus labor induction.
Higher body mass index was associated with slower labor progress and longer labor duration in patients ultimately undergoing vaginal delivery. This difference was driven by longer times spent in latent labor, with no significant differences observed in active labor. Results suggest a potential need for the use of labor curves more specifically tailored to body mass index when assessing for progress in labor, in order to avoid performance of unnecessary Cesarean deliveries.
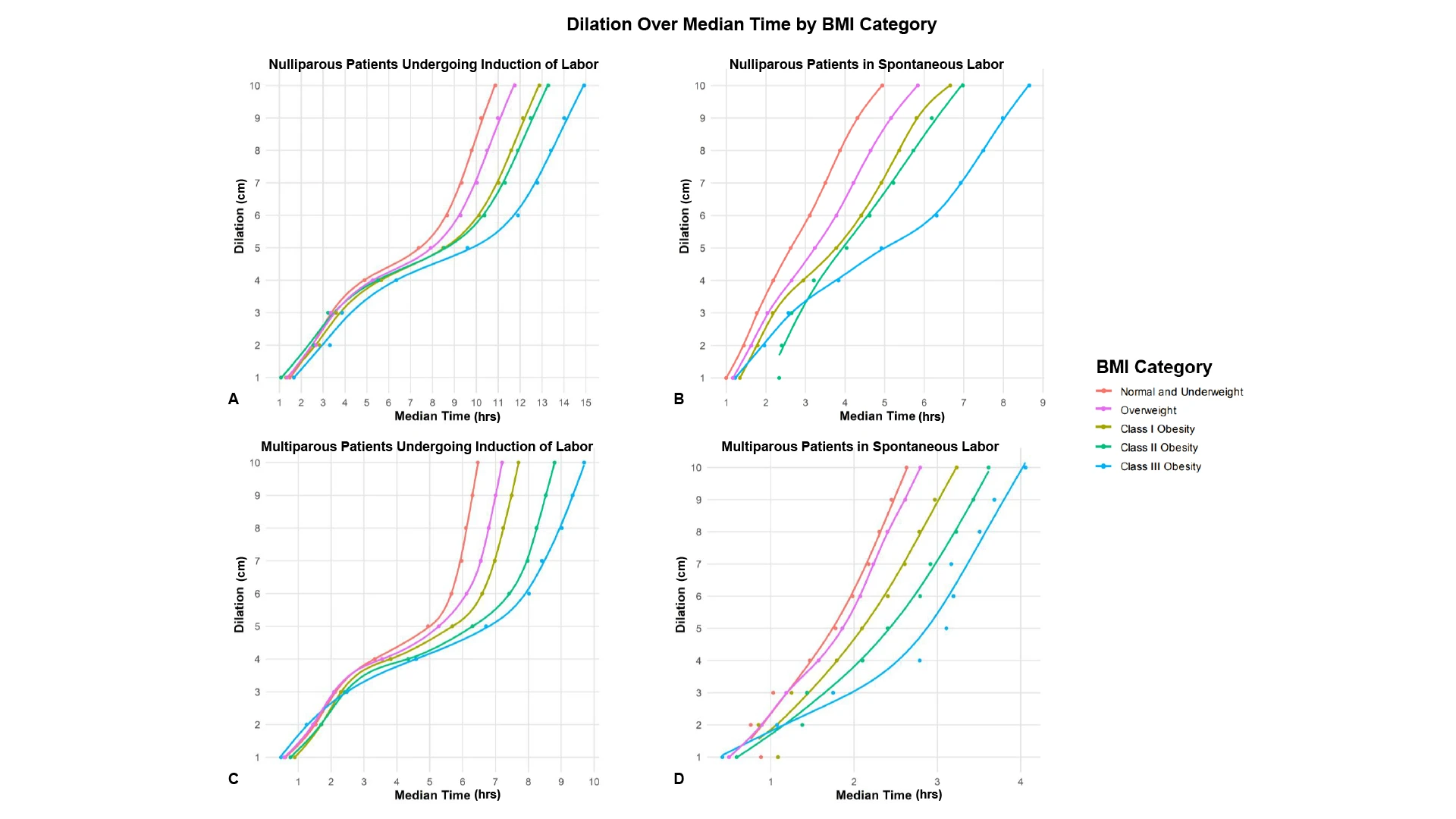
The median time in active labor did not consistently differ significantly among the BMI groups. In addition, these monotonic differences were observed when assessing various subcategories of patients, including subanalysis of nulliparous patients who underwent IOL, nulliparous patients in spontaneous labor, multiparous patients who underwent IOL, and multiparous patients in spontaneous labor. Traditionally constructed labor curves demonstrated the same patterns overall.
The Takeaway: Obesity is the most common medical condition among reproductive-aged individuals. It is also associated with increased rates of Cesarean delivery, prompting questions about avoiding potentially unnecessary Cesareans for these patients. Labor curves, which plot progression of cervical dilation over time and serve as benchmarks to assess labor progressare largely based on patients with normal BMI.
We hypothesized that among obese patients that ultimately delivered vaginally, the first stage of labor (progression to complete dilation) would be significantly longer when compared to patients with normal weight, and demonstrated this to be true. We assessed the records of all vaginal deliveries at The Mount Sinai Hospital from 2013 through 2022 and plotted out their progress via documented cervical exams, which we used to construct labor curves stratified by BMI category.
As suspected, patients with obesity, even when ultimately delivering vaginally, had significantly longer first stages of labor. Additionally, increasing obesity was associated with longer labor in a dose-dependent fashion. Our curves demonstrate that labor for patients with obesity may simply take longer, and that potentially adjusting clinical benchmarks for labor progression in these patients may avoid unnecessary Cesarean deliveries.
“Stratafix vs Vicryl suture for hysterotomy closure in scheduled Cesarean deliveries: a randomized clinical trial” American Journal of Obstetrics & Gynecology, January 2026
Nicola F. Tavella, MPH, Ayisha B. Buckley, MD, Camila Cabrera, MD Calvin E. Lambert, Jr, MD; Ariana Mills, MD, Keisha S. Paul, MS, Monica J. Patel, MPH, Luciana A. Vieira, MD, MS, Angela T. Bianco, MD, Joanne L. Stone, MD, MS
Abstract: Reducing blood loss at the time of scheduled Cesarean improves maternal outcomes. Type of suture material and closure technique may contribute to reduction in blood loss specifically related to the incision site as well as more expeditiously achieve uterine contractility. Prior publications have noted reduced closure times when using knotless barbed suture, but available evidence remains limited.
The objective was to examine whether patients whose hysterotomies were closed with Stratafix® barbed suture at the time of scheduled cesarean delivery had lower quantified blood loss compared to patients who received Vicryl® (Polyglactin 910) suture.
This was a prospective randomized clinical trial at a single tertiary care center in New York City. Patients with a scheduled primary or repeat Cesarean delivery ≥37.0 weeks’ gestation provided informed consent and were randomized to either Stratafix barbed suture or Vicryl (Polyglactin 910) suture for hysterotomy closure. The primary outcome was quantified blood loss, measured in milliliters using the mobile Stryker Triton artificial intelligence scanner. Secondary outcomes included (1) single-layer hysterotomy closure time, (2) use of additional hemostatic sutures, (3) intraoperative use of hemostatic agents, (4) rate of surgical site infection (including endometritis), and (5) differences in average postoperative pain. The Mann-Whitney U test compared observed medians of continuous outcomes between groups. Chi-squared tests compared proportional differences in categorical outcomes between groups. An a posteriori multivariable quantile regression model examined whether continuous outcomes differed when controlling for suspected confounders.
Between July 20, 2021, and November 30, 2023, 226 patients were randomized to either Stratafix barbed suture (n=113) or Vicryl (Polyglactin 910) suture (n=113) at the time of scheduled Cesarean delivery. We observed no significant difference between the median quantified blood loss observed in the Stratafix barbed suture group (544.5 [250.7–838.8] mL) and the Vicryl (Polyglactin 910) suture group (600 [348–852] mL; P=.22). Median time to hysterotomy closure was 1.3 minutes shorter in the Stratafix barbed suture group (4.1[2.6–5.6] minutes) and the Vicryl (Polyglactin 910) suture group (5.4 [3.5–7.2] minutes; P<.001). In an a posteriori multivariable quantile regression adjusting for confounders, the median total quantified blood loss was not statistically significantly different between groups (P=.37). Median time to hysterotomy closure was 1.8 minutes shorter for the Stratafix barbed suture group (P<.01).
The use of Stratafix barbed sutures was not associated with a reduction in blood loss compared with the Vicryl (Polyglactin 910) suture. However, the results of this randomized clinical trial suggest that Stratafix barbed suture may be associated with faster single-layer hysterotomy closure time. This suggests that Stratafix barbed suture could have benefits over Vicryl (Polyglactin 910) suture for the single-layer closure of hysterotomy during cesarean delivery. Any possible benefits of using Stratafix barbed suture at the time of hysterotomy closure during scheduled cesarean require further exploration.
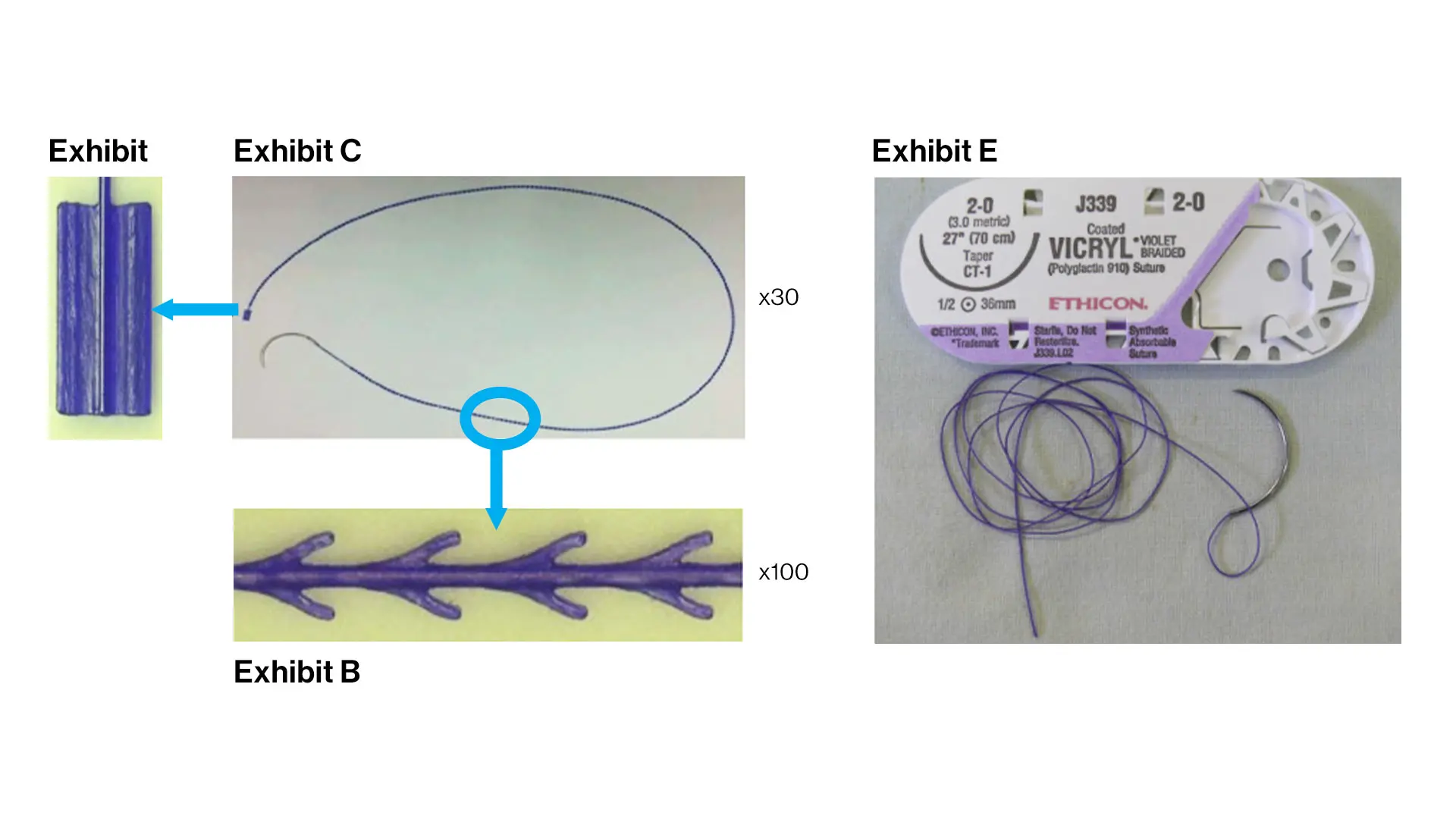
The Stratafix® barbed suture, left, and the Vicryl® (Polyglactin 910) suture.
The Takeaway: This randomized clinical trial helps clarify the role of knotless barbed suture for hysterotomy closure at scheduled Cesarean delivery. While Stratafix did not significantly reduce quantified blood loss compared with Vicryl, it consistently shortened single-layer closure time by approximately one-two minutes. In the context of Cesarean delivery—where efficiency, reproducibility, and surgeon ergonomics matter—this finding is meaningful. Faster closure may translate to smoother operative flow and potentially less uterine manipulation, even if it does not directly reduce blood loss in low-risk, scheduled cases.
Importantly, the absence of a blood-loss benefit suggests that suture choice alone may not be sufficient to influence hemorrhage outcomes in this population, where baseline risk is relatively low and hemostasis is multifactorial. However, operative speed and tissue handling may have implications beyond the index surgery.
The next logical step is to move beyond short-term intraoperative outcomes and examine uterine healing. A planned follow-up study evaluating postoperative myometrial thickness in patients randomized to barbed versus conventional suture would address a clinically relevant question in more intuitive terms: does faster, more evenly distributed closure lead to a thicker, potentially stronger uterine scar? This work could help determine whether the advantages of barbed suture extend to longer-term structural outcomes that matter for future pregnancies and surgical decision-making.
“Intrahepatic Cholestasis of Pregnancy Recurrence in a Subsequent Pregnancy,” Obstetrics and Gynecology, August 2025.
Henri M. Rosenberg, MD, Minhazur R. Sarker, MD, Gladys A. Ramos, MD, Angela Bianco, MD, Lauren Ferrara, MD, Chelsea A. DeBolt, MD.
Abstract: Reported recurrence rates of intrahepatic cholestasis of pregnancy (ICP) range from 40 percent to 90 percent based on prior literature, but data remain limited. In a retrospective cohort, we aimed to evaluate the rate of ICP recurrence and risk factors associated with recurrence. Among 104 patients with ICP in an index pregnancy, 46 experienced recurrence (44 percent) of ICP in subsequent pregnancy. A peak total bile acid (TBA) level greater than 40 micromoles/L in the index pregnancy was significantly associated with ICP recurrence; shorter interpregnancy intervals were associated with lower risk. These findings contribute to the literature on the risk of ICP recurrence in a modern, racially diverse cohort and highlight elevated TBA level as a predictor of recurrence.
Intrahepatic cholestasis of pregnancy (ICP) is the most common hepatobiliary disease of pregnancy and is associated with significant perinatal morbidity and mortality. The etiology is considered multifactorial, influenced by hormonal, environmental, and genetic factors. Prior studies estimate an ICP recurrence rate after an affected pregnancy of 40–90 percent. However, these studies were completed in predominantly white European and Asian populations, reducing generalizability. Our objective was to estimate the recurrence of ICP in subsequent pregnancy and identify risk factors associated with ICP recurrence in a modern, racially diverse population.
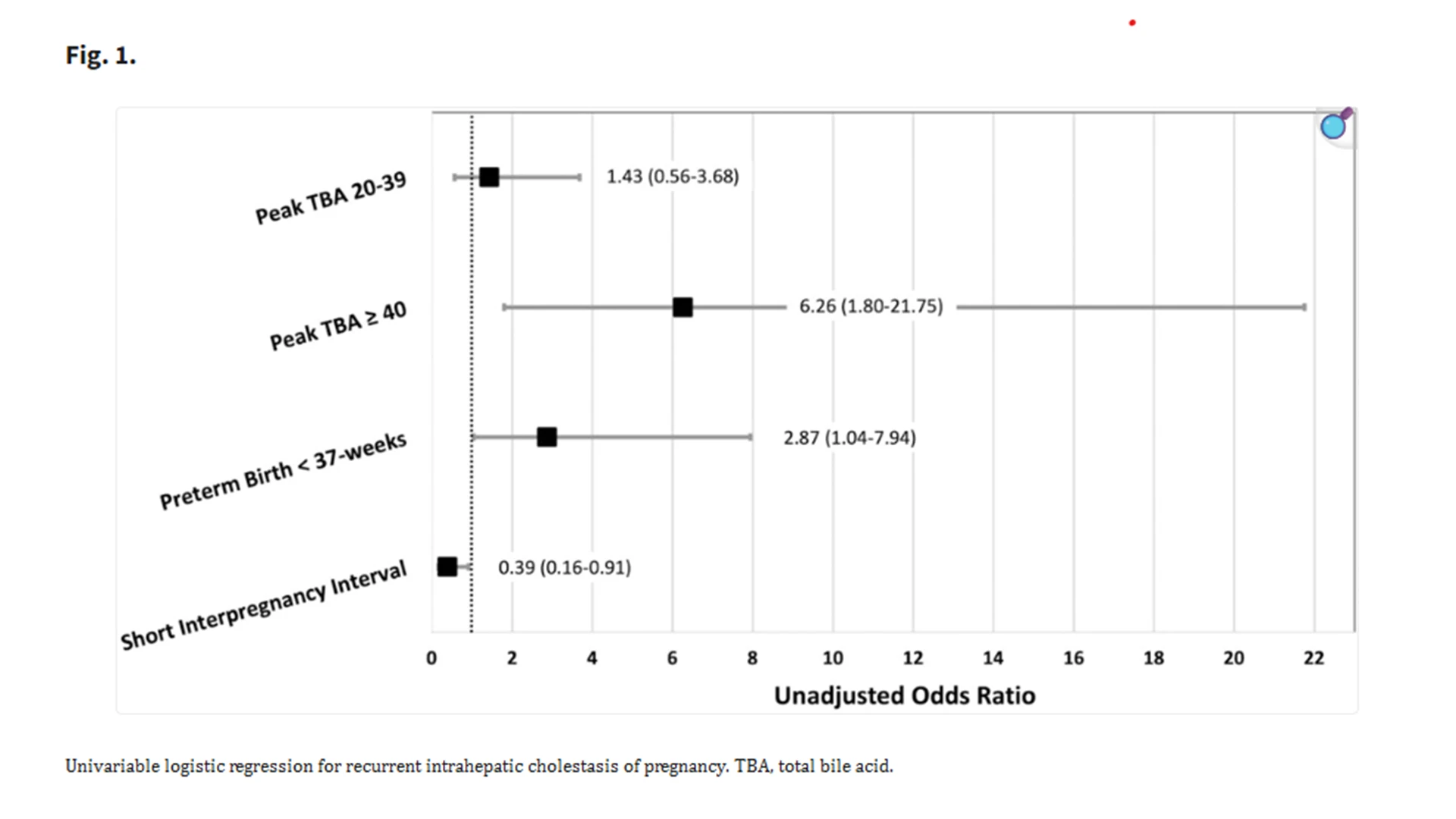
A short interpregnancy interval (defined as time from delivery to next conception less than 18 months) was associated with decreased odds of ICP recurrence.
The Takeaway: Intrahepatic cholestasis of pregnancy remains one of the most clinically challenging hepatobiliary disorders of pregnancy because of its association with stillbirth, spontaneous preterm birth, and medically indicated early delivery. Prior recurrence estimates—ranging from 40–90 percent—were drawn largely from older European and Asian cohorts and frequently relied on patient-reported symptoms rather than biochemical confirmation. Consequently, clinicians have lacked contemporary, U.S.-based data drawn from racially and ethnically diverse patient populations that better reflect real-world clinical practice.
Our study addressed this gap by evaluating recurrence in a modern, diverse U.S. cohort using strict biochemical criteria.
We identified a recurrence rate of 44 percent, but the most important insight was that disease severity in the index pregnancy strongly predicted recurrence. Patients with peak total bile acid concentrations exceeding 40 micromoles/L had markedly higher recurrence risk, suggesting that underlying susceptibility in bile acid synthesis or transport pathways may persist across pregnancies.
These findings allow for more precise anticipatory guidance. Patients with significant bile acid elevation may benefit from earlier symptom assessment, earlier bile acid testing, and more proactive management strategies in future pregnancies. Conversely, those with milder disease may have a lower recurrence risk than previously believed.
Our next steps include validating these predictors in larger, multicenter cohorts with broader racial and ethnic representation. We also aim to integrate genomic data to determine whether variants in bile acid transport pathways interact with hormonal or environmental factors to shape recurrence. Ultimately, our goal is to develop individualized recurrence prediction tools that incorporate biochemical, genetic, and clinical factors to support more personalized counseling and targeted surveillance in subsequent pregnancies.
“Unlocking the therapeutic potential of rigosertib as a selective therapy for ovarian cancer” Cell Reports Medicine, July 2025.
Shalini Nath, PhD, Sally Claridge, PhD, Genesis Lara Granados, Majd Al Assaad, MD, Eric Park, Julie-Ann Cavallo, PhD, Ufuoma Nuwere, Maame Esi Ackon, Lamberto De Boni, Stacey Baker, E. Premkumar Reddy, PhD, Stephanie V. Blank, MD, Olivier Elemento, Rachel Brody, MD, PhD, Benjamin D. Hopkins, PhD
Abstract: Precision oncology seeks to exploit tumor-specific drug sensitivities. Traditionally, this is accomplished through the identification and targeting of highly recurrent mutations. This paradigm falls short in ovarian cancer, where the oncogenic alterations are more diverse, necessitating an alternate approach for the identification of tumor-specific vulnerabilities. To address this, we have used a functional modeling approach, integrating drug screening with a Kinome Atlas-based assessment of signaling, to nominate a therapeutic regimen for ovarian tumors. This approach identifies a small-molecule RAS mimetic, rigosertib, as a tumor-selective agent and leads us to identify the combination of rigosertib with phosphoinositide 3-kinase (PI3K) or mammalian target of rapamycin (mTOR) inhibition as effective combinations that prevent rigosertib-induced survival signaling while inducing regressions in ovarian cancer xenografts. These data support further exploration of these combinations for the treatment of ovarian cancer.
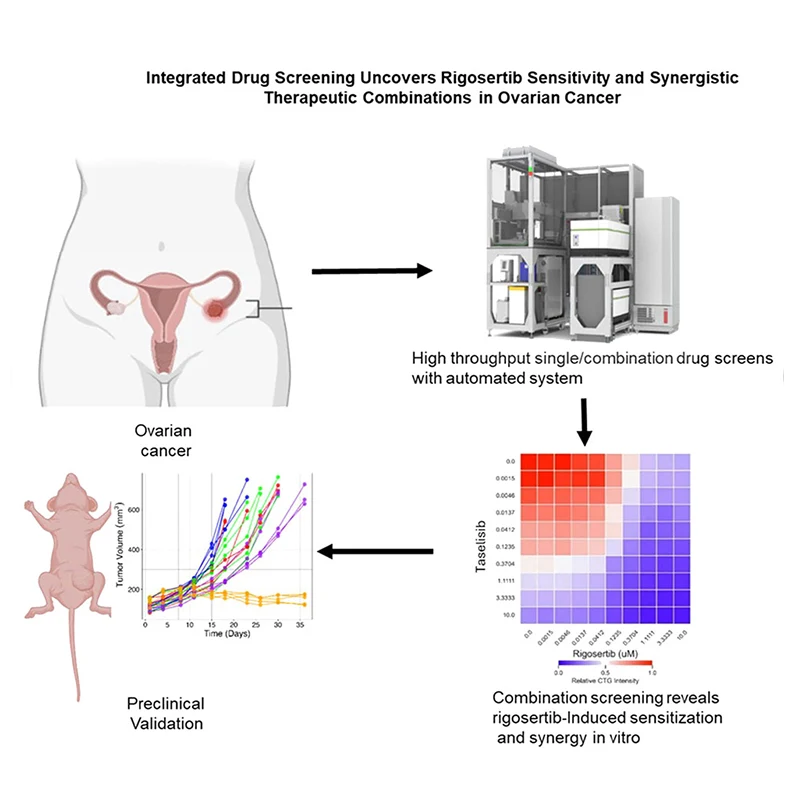
A graphical abstract of the publication.
The Takeaway: Ovarian cancer remains one of the deadliest cancers, with most patients eventually developing resistance to standard chemotherapy. The current study used an innovative approach to find better treatments by testing 117 drugs across multiple cancer types to identify therapies specifically effective against ovarian cancer. The study identified rigosertib, a drug that blocks a protein called RAS, as a potentially active therapeutic strategy in ovarian cancer. However, treatment with rigosertib activated a compensatory signaling through the PI3K/mTOR signaling pathway, which limited the drug efficacy. To overcome this, the investigators used combination of rigosertib with drugs targeting the PI3K pathway. This combination proved much more effective than either drug alone, significantly shrinking tumors in mice without causing serious side effects.
Importantly, this combination outperformed cisplatin, the current standard chemotherapy, in laboratory studies. The findings suggest that blocking both growth pathways simultaneously may offer a promising new treatment strategy for ovarian cancer patients, particularly those whose cancer has become resistant to conventional therapies. Clinical trials will be needed to test this approach in patients.
“Unnecessary exclusions: eligibility criteria in gynecologic oncology interventional clinical trials impairs access,” International Journal of Gynecologic Cancer, May 2025.
Sharonne Holtzman, MD, Riva Letchinger, MD, Lily McCarthy, MD, Isabel S. Chess, MD; Daniel Liu, MD, Sunidhi Singh, MD, Dmitry Zamarin, MD, PhD, Konstatin Zakashansky, MD, and Stephanie V. Blank, MD
Abstract: This study aimed to assess the eligibility requirements in gynecologic oncology clinical trials that may impair patient access to clinical trials.
Using clinicaltrials.gov, gynecologic oncology interventional studies conducted between September 1, 1997, and September 1, 2023, were surveyed. Studies were included if they were interventional and conducted in the United States, with available protocols. Differences in means were estimated, and hypothesis testing was conducted under the general framework for bimodal logistic regression or paired t tests where appropriate.
Of the 606 included interventional clinical trials, 256 (42.2 percent) were for uterine cancer, 99 (16.3 percent) for ovarian, fallopian tube or peritoneal cancer, 55 (9.1 percent) for cervical cancer, and 197 (32.5 percent) involved multiple gynecologic oncology cancers. Of all eligible clinical trials, 351 (57.9 percent) had an exclusion criterion based on renal function, 206 (34.0 percent) had a diagnosis of human immunodeficiency virus, 194 (32 percent) had a mental health or psychiatric condition, and 170 (28.1 percent) had an exclusion criterion based on investigator discretion. Renal exclusion was more likely in clinical trials for uterine cancer (p = .04). Exclusion based on investigator decision was more likely in uterine cancer (p = .001) and ovarian cancer trials (p = .007) than in cervical cancer trials.
The most frequent exclusion criteria in gynecologic oncology clinical trials were based on renal function, followed by diagnosis of human immunodeficiency virus status and diagnosis of psychological/mental illness. Our study emphasizes the importance of understanding eligibility requirements of clinical trials to increase access to clinical trials for all patients.
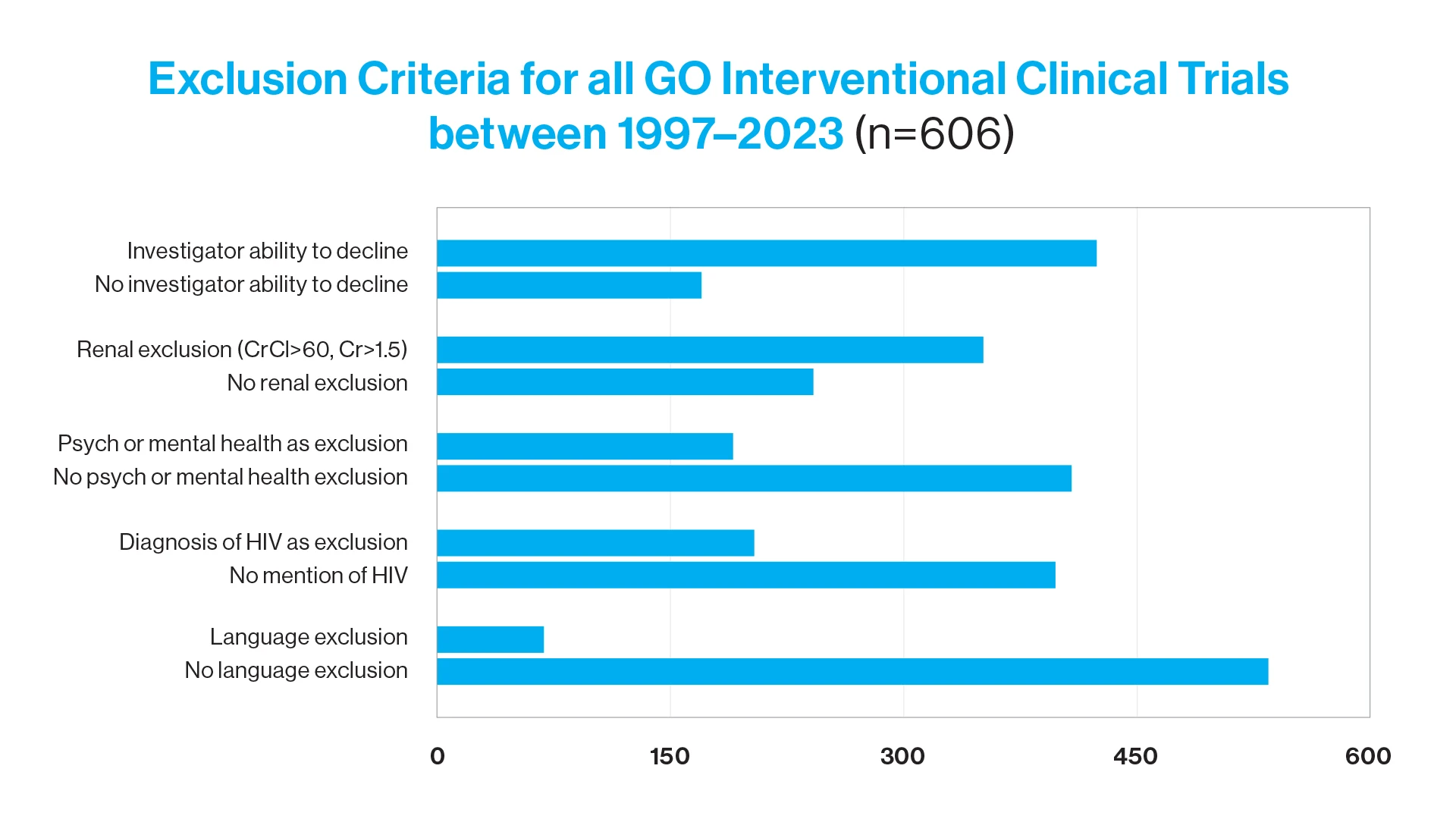
Exclusion criteria for all GO interventional clinical trials between 1997 and 2023 (n = 606). Cr, Creatinine; CrCI, Creatinine Clearance; GO, gynecologic oncology; HIV, human immunodeficiency virus.
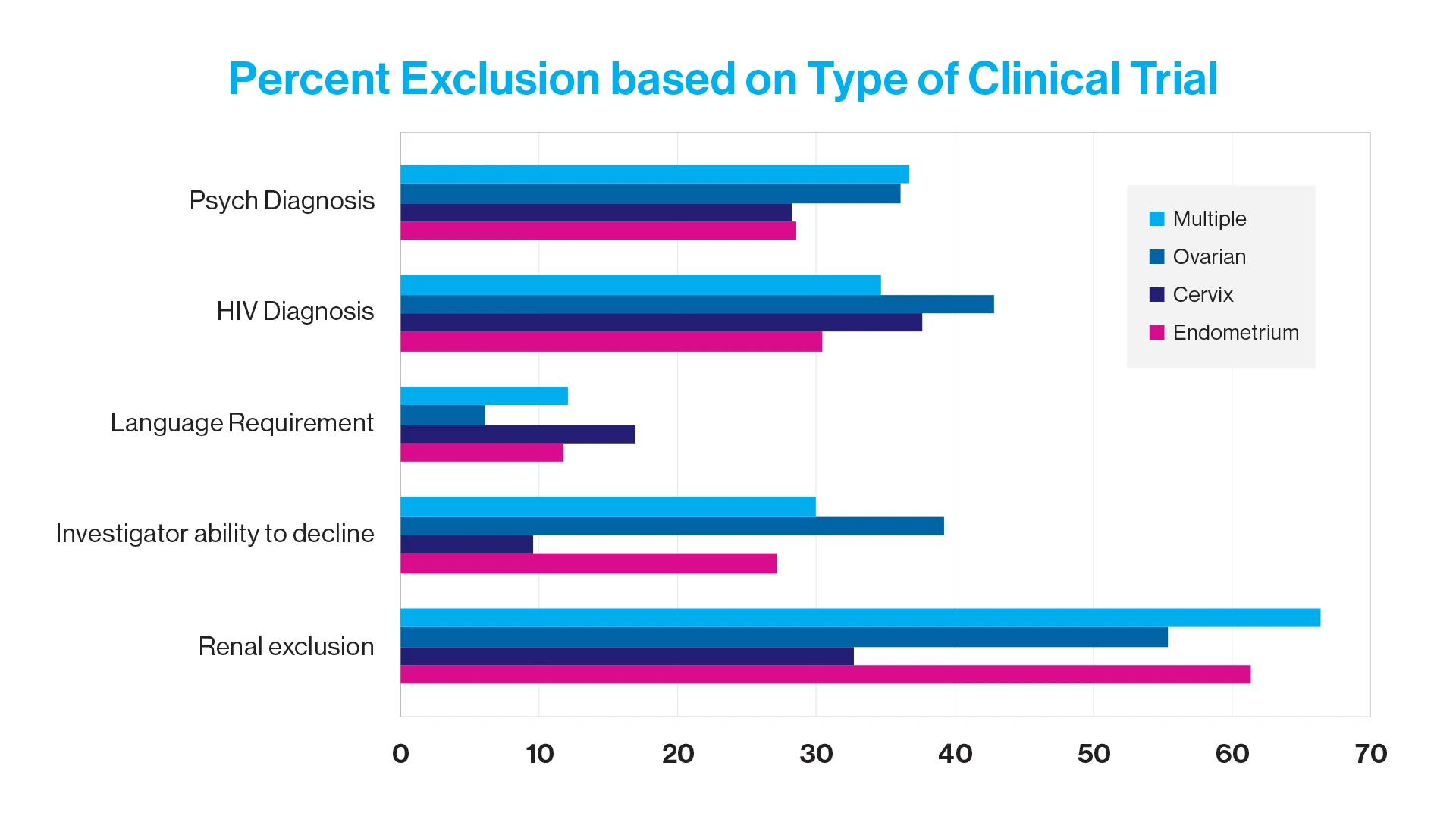
Percent exclusion based on type of clinical trial. HIV, human immunodeficiency virus.
The Takeaway: This study is significant because it highlights a persistent and under-recognized issue in clinical trials for oncology and specifically in gynecologic oncology. Clinical trials are intended to produce evidence that can be applied broadly, yet overly narrow inclusion/exclusion criteria can skew the study cohort toward younger or healthier patients, reducing external validity and leaving clinicians uncertain about how results apply to real-world patients. By systematically characterizing these criteria and quantifying their potential impact on trial representativeness, we provided technical evidence that current trial design practices may inadvertently perpetuate disparities and reduce clinical applicability of clinical trials to our patient population.