Even with the benefit of an effective vaccine and periodic screening, hepatitis B virus (HBV) remains a global scourge, the leading cause of hepatocellular carcinoma and more than a million liver-related deaths each year.
Mount Sinai Health System has taken a leadership role in battling this public health menace, launching a universal screening and vaccination campaign against the highly contagious virus across New York City in the fall of 2022, while heading up an international team of hepatologists and infectious disease experts who recently issued a bold new set of guidelines to streamline testing, diagnosis, and management of hepatitis B as a way to achieve the World Health Organization (WHO) goal of eliminating HBV.
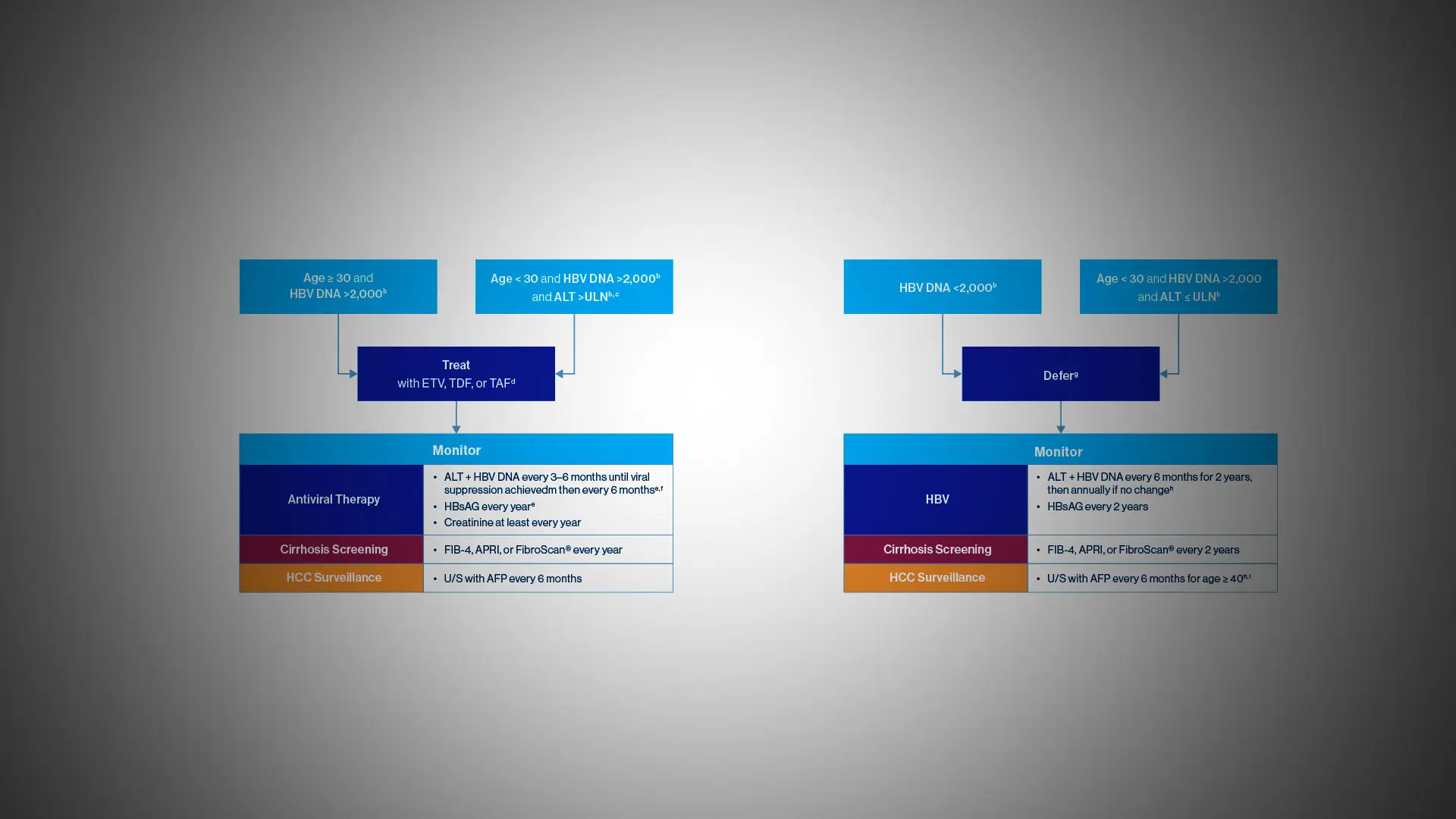
“Current guidelines for screening, vaccination, and treatment are overly complex and have left too many people undiagnosed and improperly managed,” says Douglas Dieterich, MD, Director of the Institute for Liver Medicine for the Mount Sinai Health System, and lead author of a study in the journal Gastro Hep Advances describing the new protocols. “Our global expert panel developed a treatment algorithm to provide a streamlined visual pathway for risk stratification and management of patients with HBV that can be adapted in various care settings around the world, particularly in primary care physician offices.”
In recommending that all adults over age 18 be screened for hepatitis B at least once in their lifetime, the team of experts broke sharply with medical society and Centers for Disease Control and Prevention (CDC) guidance that testing only be done for people at elevated risk. Moreover, the panel recommended, for the first time, HBV screening for all pregnant women with each pregnancy, as well as broad testing for coinfections, including hepatitis C virus, human immunodeficiency virus, and hepatitis delta virus (HDV).
Significantly, the new guidelines coincide with the release in March 2023 of updated recommendations from the CDC for minimum once-in-a-lifetime adult screening through a triple panel test for hepatitis B surface antigen (HBsAg), antibody to hepatitis B surface antigen (anti-HBs), and total antibody to hepatitis B core antigen (total anti-HBc). Additional guidance released in 2022 from the CDC’s Advisory Committee on Immunization Practices (ACIP) urging hepatitis B vaccination for all adults ages 19 to 59 (and those over 60 with risk factors) also mirrors recommendations from the Mount Sinai-led task force for universal vaccinations against HBV in all patients who have negative serologies, given the fact it is impossible to determine potential future risks for exposure to HBV.
The simplified approach was hailed in an editorial accompanying the study as “a bold and important expansion of treatment eligibility” and “a necessary policy change given the flawed and failed approach to risk-based screening, and [one that] is likely to be cost-effective.”
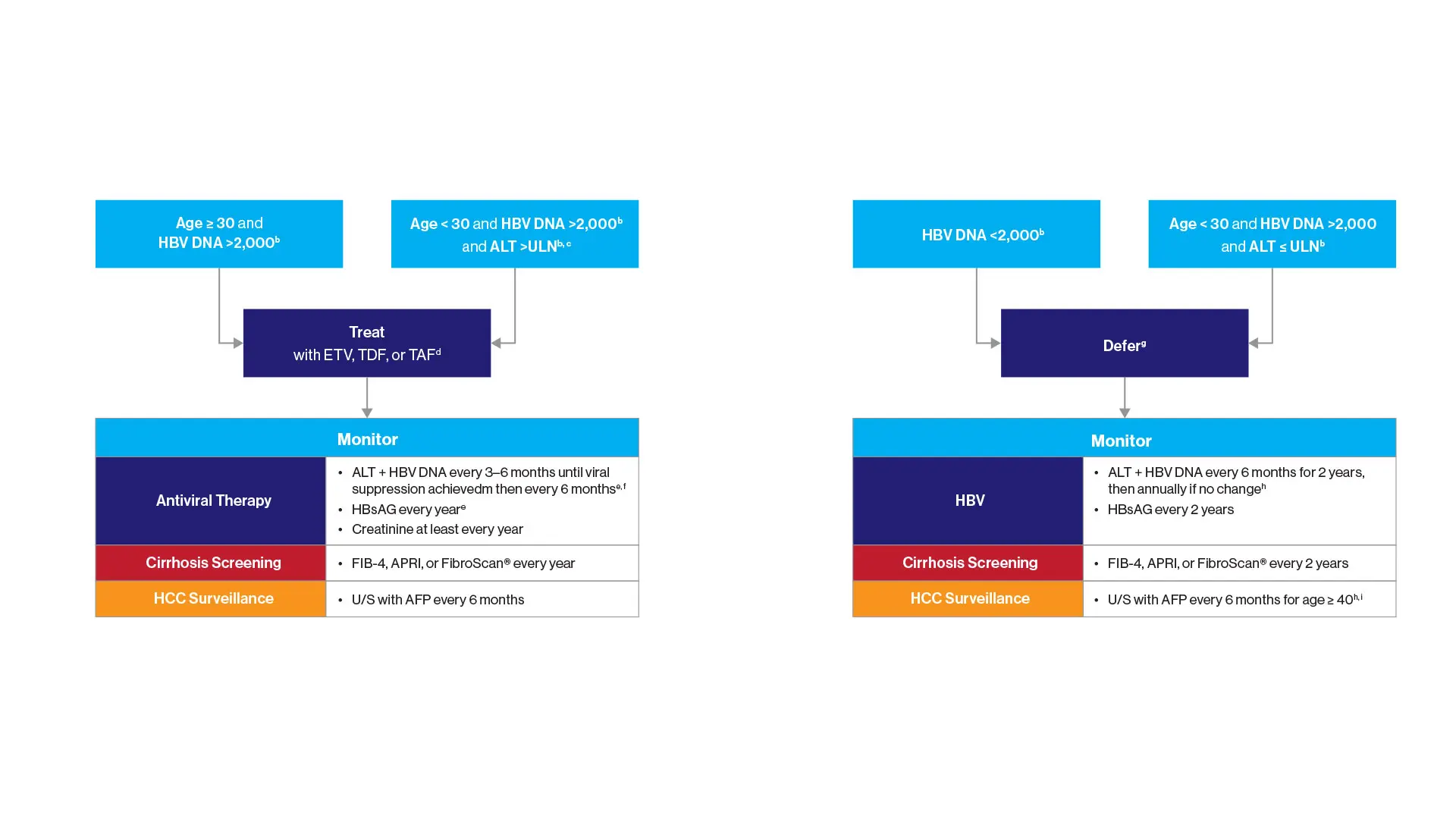
Guidelines developed by Mount Sinai researchers for hepatitis B testing, diagnosis, and management
In issuing its own updated guidance, the CDC noted that far too many people with hepatitis B in the United States are unaware they carry the infection, each representing “a missed opportunity for preventing morbidity and mortality.” The best way to address that health care gap, the CDC further stated, is through universal health care screening in concert with more aggressive hepatitis B vaccination. Five vaccines are currently approved by the U.S. Food and Drug Administration for use in adults: PreHevbrio, Recombivax HB, Engerix-B, TWINRIX (a hepatitis A and hepatitis B combination), and Heplislav-B (two doses one month apart, compared to three doses for the other vaccines).
In the face of public health evidence that New York City has an estimated 243,000 individuals with chronic hepatitis B—of whom an estimated 46 percent are undiagnosed—Mount Sinai has undertaken its own universal surveillance and vaccination initiatives, piggybacking on a program targeting hepatitis C that was rolled out in 2015 and expanded since then. The latest effort has prompted a dramatic increase since last fall in the numbers of patients being screened for HBV, according to Dr. Dieterich, who oversees liver care across the Mount Sinai Health System.
Helping to drive that upsurge are the thousands of hepatitis B surface antigen-positive patients identified from Mount Sinai’s vast electronic medical records; these patients are then encouraged to seek treatment through the Institute for Liver Medicine. Another driver is a novel program of medical alerts that appear on the screens of primary care physicians when a patient in their office has no record of hep B or hep C serology results. In these cases, the doctors can simply click on their screens to order the appropriate tests for their patients.
Mount Sinai’s campaign also features strong public awareness and community outreach components. The former is being spearheaded by an expanding program of social media posts (Twitter, Facebook, LinkedIn, Instagram), and the latter by multilingual health care coordinators who visit neighborhoods throughout the city to educate people about hepatitis viruses, while setting up large-scale screening events.
“We’re committed to the goals of the World Health Organization to eliminate hepatitis B from the planet through active and aggressive surveillance,” emphasizes Dr. Dieterich, “and we’re very pleased to say our efforts are having an impact. We’re bringing growing numbers of patients into the treatment fold before they progress to more severe forms of the deadly disease.”
Featured

Douglas Dieterich, MD
Professor of Medicine (Liver Diseases); Director, Institute for Liver Medicine

Meena Bansal, MD
Professor of Medicine (Liver Diseases); System Chief, Division of Liver Diseases