Urinary incontinence has long been a common complication among patients who have undergone robot-assisted radical prostatectomy (RARP). A novel surgical technique developed at Mount Sinai is showing considerable promise in facilitating early return of continence with low residual cancer rates.
Developed by Ash Tewari, MBBS, MCh, Professor and Chair, Milton and Carroll Petrie Department of Urology, and the Kyung Hyun Kim, MD, System Chair in Urology, the “hood technique” is unique in that it uses an anterior approach to preserve the contents of the space of Retzius during robot-assisted radical prostatectomy.
Upon removal of the prostate, the preserved tissue has a “hood-like” appearance, comprised of the detrusor apron, arcus tendineus, puboprostatic ligament, anterior vessels, and some fibers of the detrusor muscle. The hood effectively encloses and protects the membranous urethra, external sphincter, and supportive structures.
A previously developed anterior-posterior technique in which the prostate and the bladder neck are approached through the pouch of Douglas was shown to be effective in preserving the Retzius space and its contents and facilitating an early return to continence among patients following RARP. However, the hood technique recognizes that many surgeons perform robotic prostatectomy from the anterior aspect.
“Using our technique, surgeons are not only able to preserve these anterior structures that are crucial to restoring early continence following radical prostatectomy but also do so with low positive surgical margin rates and enhanced ability to visualize anatomical landmarks,” Dr. Tewari says. “That is what makes our technique so unique.”
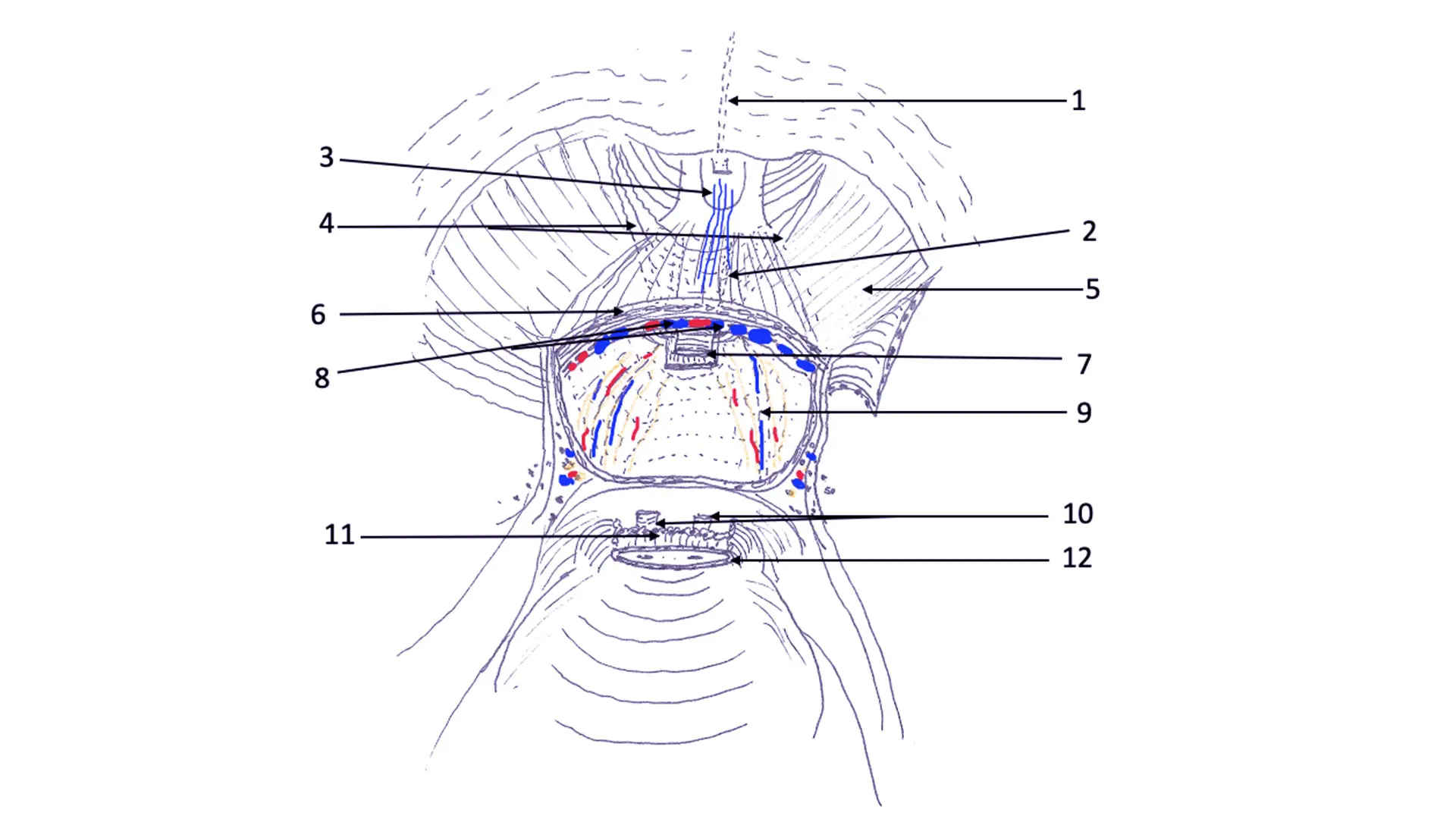
Sketch demonstrating Hood surgical anatomy. Anatomical components of Hood surrounds and safeguards membranous urethra and external urethral sphincter and thereby urethrovesical anastomosis.
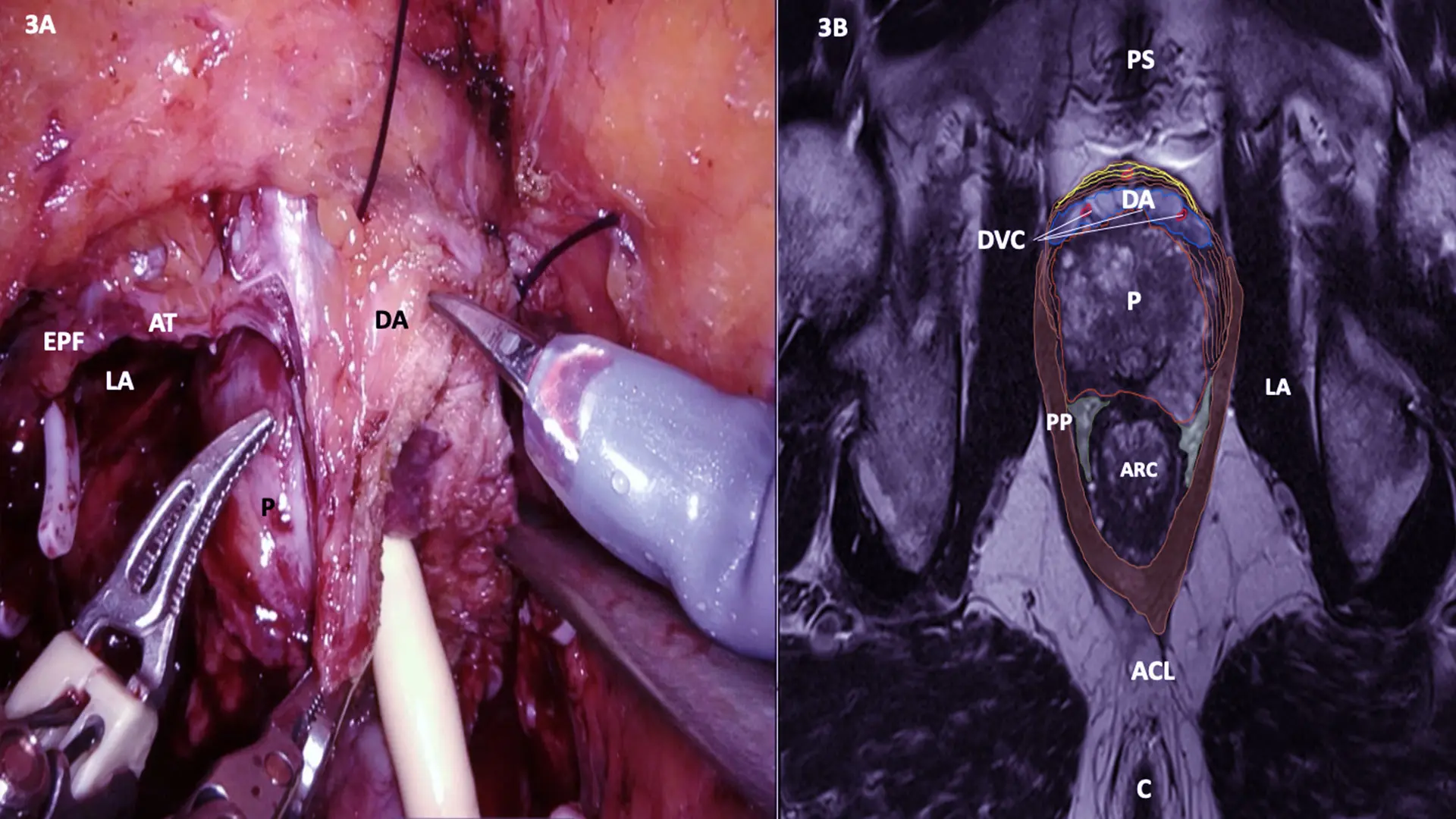
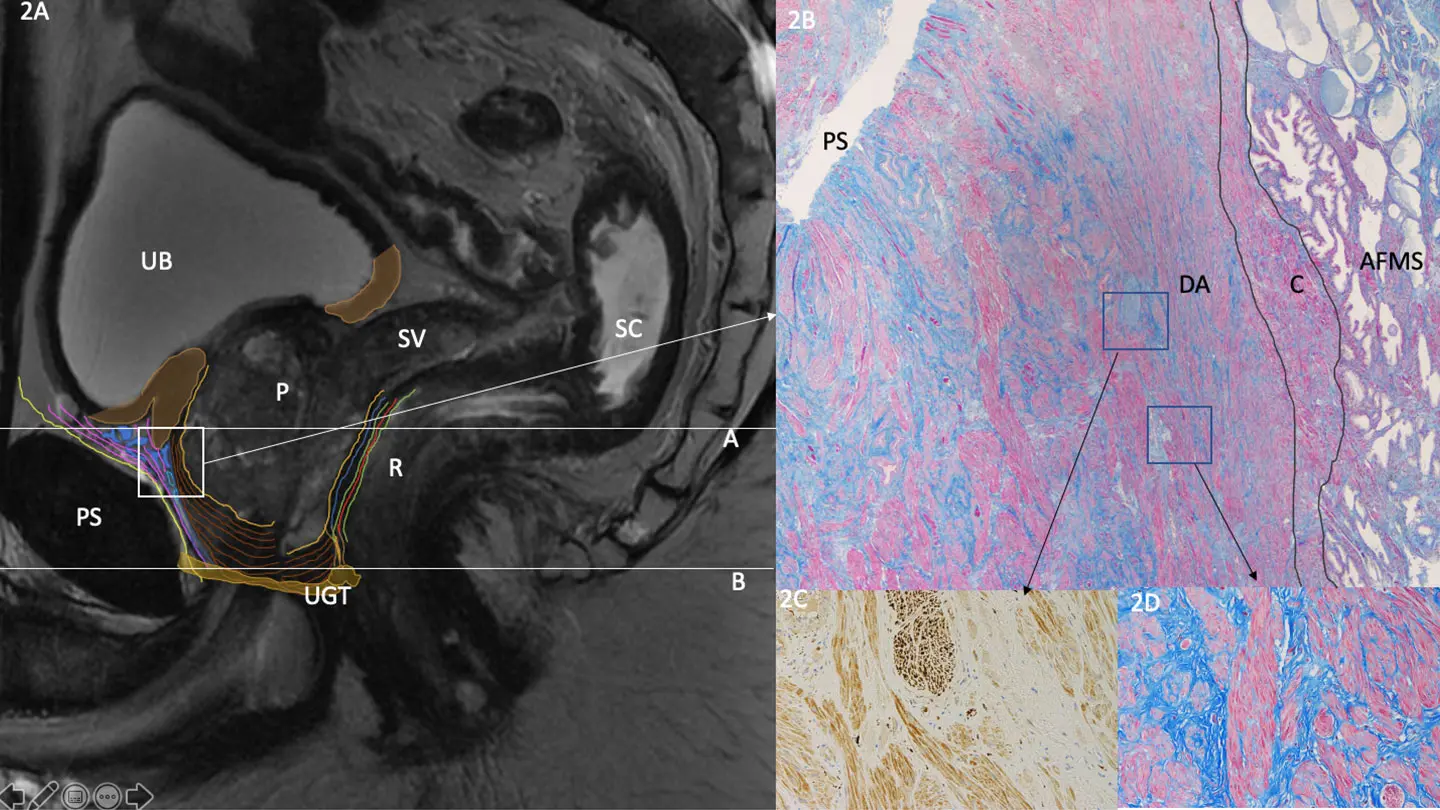
MRI Cross section at A line with Surgery still Image
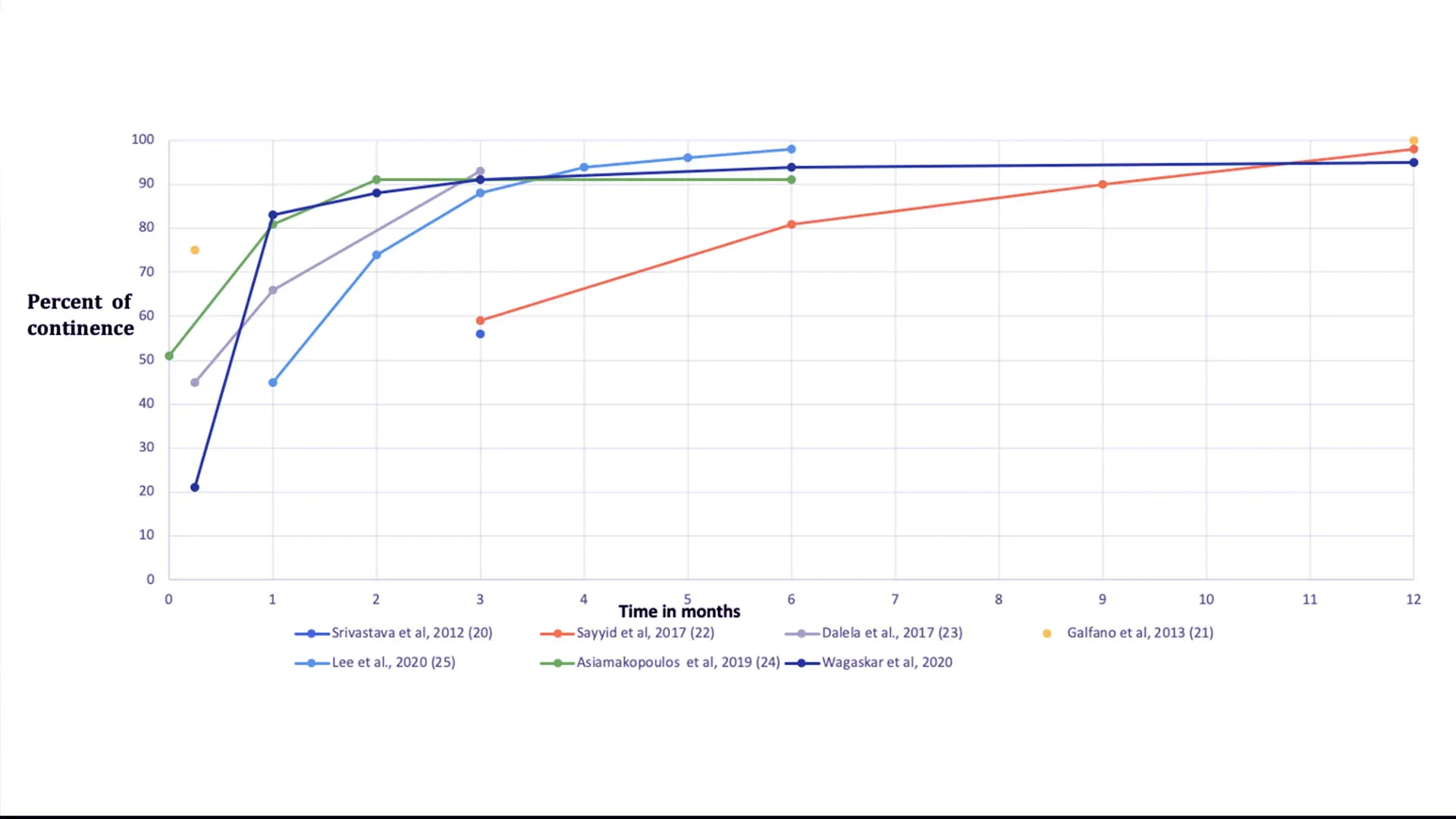
Comparison of continence rates between current study, previous study from the same group, and published Retzius-sparing approach.
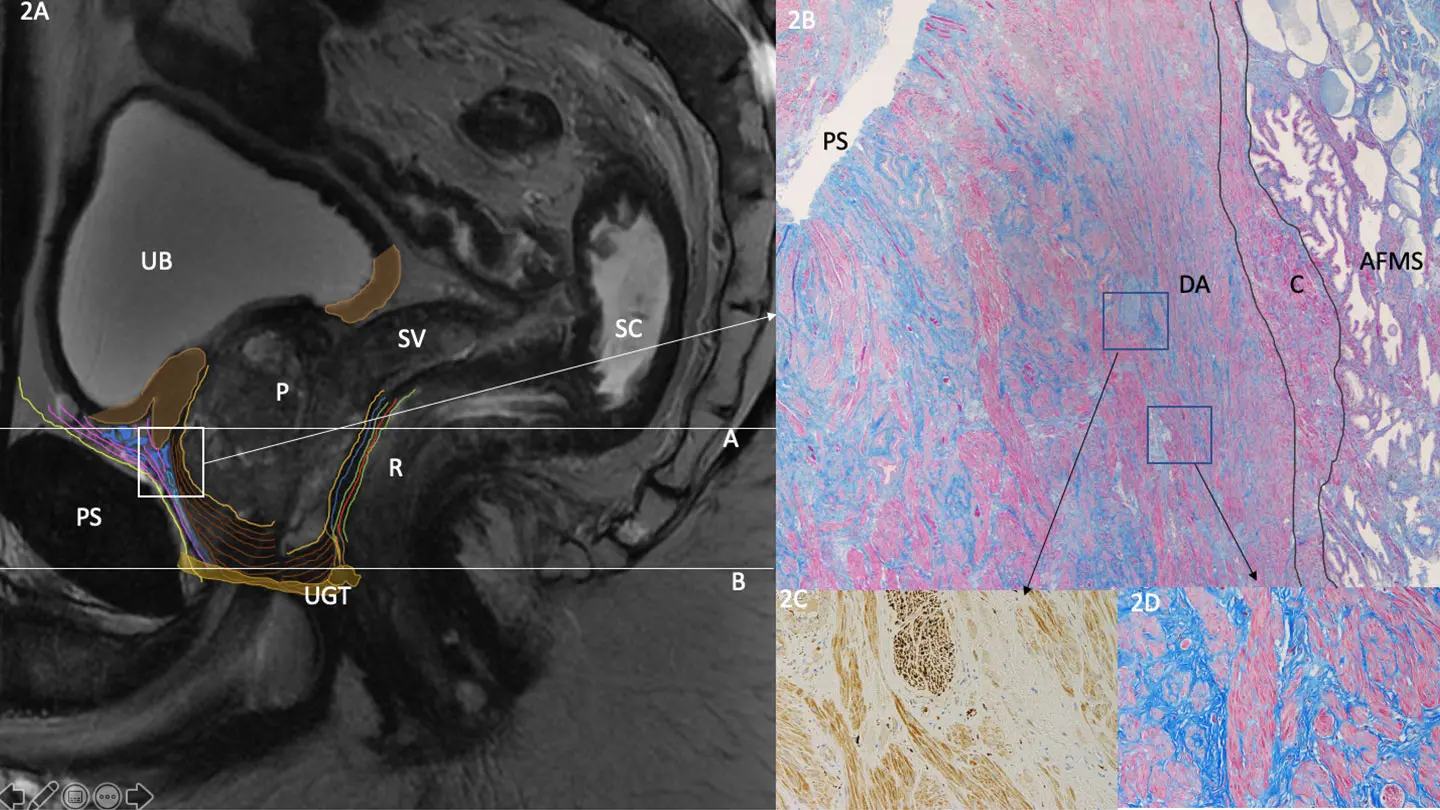
Sagittal Pelvic MRI with Detrusor Apron Histology
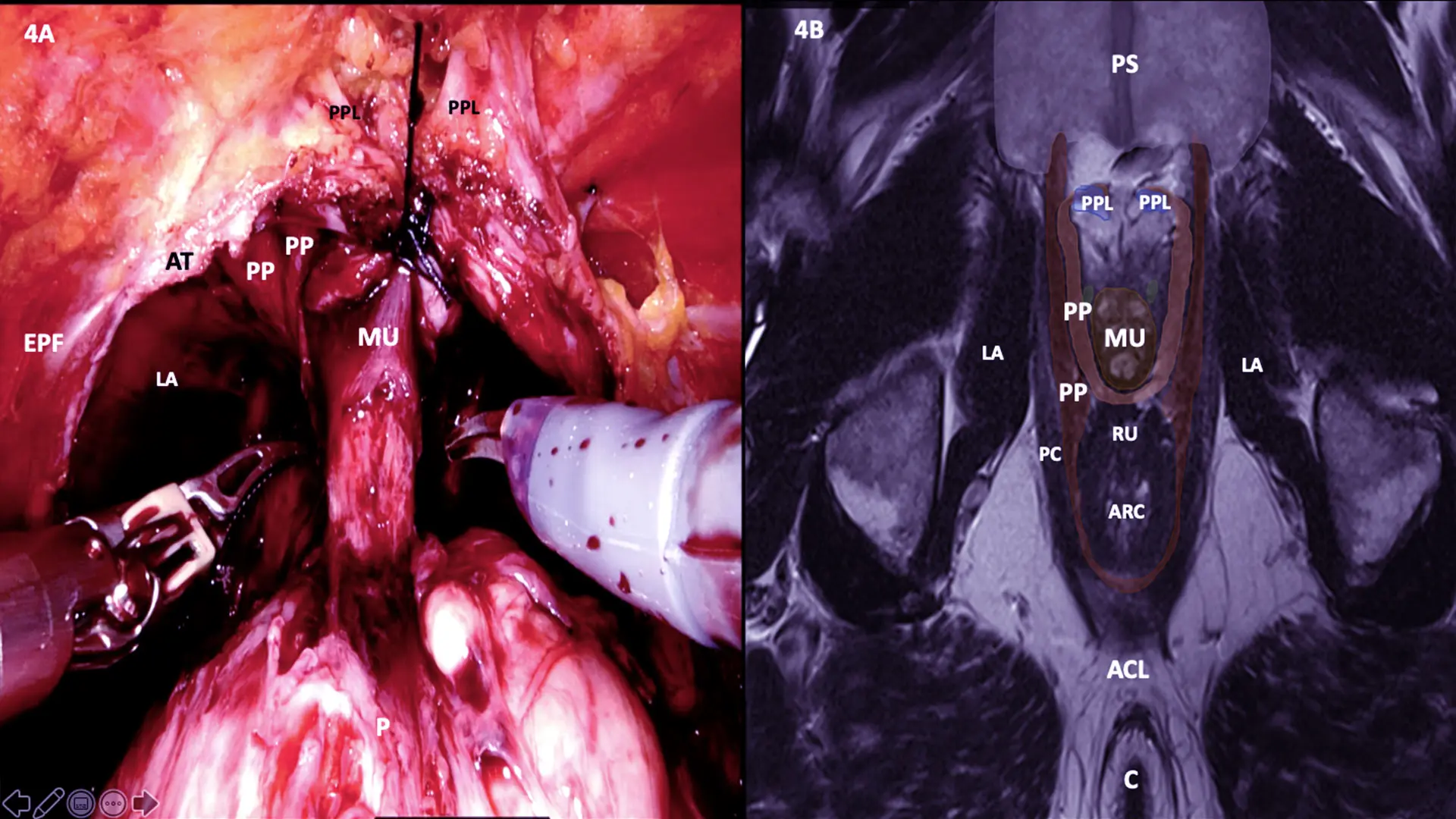
MRI Cross-section at B line with Surgery still image
To assess the efficacy of RARP with the hood technique, Dr. Tewari conducted a prospective study of 300 patients (median age 64) with localized prostate cancer between April 2018 and March 2019. Patients with anterior tumor location based on biopsy or multiparametric magnetic resonance imaging were excluded as were those who were being administered hormone or radiation therapy.
Pelvic lymph node dissection (PLND) was performed among patients with Gleason score 3 + 4, with limited PLND performed among Gleason score 7 patients, and extended PLND performed among patients with Gleason score 4 + 4. The median duration of the entire procedure was 169 minutes, and the median console time was 118.5 minutes, with no intraoperative complications observed among patients. The overall complication rate with the hood technique was 9.7 percent, which was within the range of published rates (4.3 percent to 19.4 percent) following RARP.
Patients were monitored for one year, initially through phone interviews and then consultations at week 6, week 12, week 24, week 36, and week 48. Of the 300 patients who underwent RARP with the hood technique, 62 (21 percent) achieved continence, defined as completely pad free, one week after catheter removal. Continence rates continued to improve among patients at week 2 (109, 36 percent), week 4 (249, 83 percent), week 6 (265, 88 percent), week 12 (274, 91 percent), week 24 (282, 94 percent), and week 48 (286, 95 percent).
At the end of the 12-month follow-up, 14 patients had not achieved continence, with nine using an occasional one pad per day (PPD), three patients using two PPDs, and one patient using four PPDs. Four of the patients using two or more PPDs developed urinary tract infections after prostatectomy, three of whom underwent biofeedback at 12 weeks following the procedure.
The study, “Hood Technique for Robotic Radical Prostatectomy—Preserving Periurethral Anatomical Structures in the Space of Retzius and Sparing the Pouch of Douglas, Enabling Early Return of Continence Without Compromising Surgical Margin Rates,” was published October of 2020 in European Urology.
“These results confirm that, by preserving anterior structures, we can achieve early continence among our patients, which is beneficial from a quality of life perspective because that means there is no requirement to use pads,” Dr. Tewari says.
Dr. Tewari also noted that the positive surgical margin rate among participants was 6 percent, which includes final pathology as well as frozen section margins for all stages. This outcome was significantly lower than the rates reported in previous Retzius-sparing technique studies, which averaged 11 percent to 25 percent for pT2 cancers and 36 percent to 47 percent for pT3 cancers.
“Another major benefit of this technique is we were able to avoid the residual cancer typically associated with achieving early continence among patients, and thus spare them the subsequent hormone or radiation treatments that compromise quality of life,” Dr. Tewari says. “With our technique, we demonstrated that it is possible to achieve early continence and remove the cancer, which is the best possible treatment we can offer.”
Given that this was a single-center, single-surgeon study, and given Dr. Tewari’s extensive experience performing more than 7,000 RARPs, the results may not be replicable by other surgeons performing the technique. Moreover, the lack of control arm necessitates verification with a randomized clinical trial. In the meantime, Dr. Tewari is proceeding with additional studies, including efficacy of the technique in restoring sexual function, and promoting the technique to urologists around the world through webinars.
“We believe the favorable functional outcomes we have achieved combined with the low rate of positive surgical margins make this technique ideal for patients with localized prostate cancer,” he says.
Featured

Ash Tewari, MBBS, MCh, FRCS (Hon.)
Professor and System Chair, Milton and Carroll Petrie Department of Urology, Director of the Center of Excellence for Prostate Cancer at The Tisch Cancer Institute at the Icahn School of Medicine at Mount Sinai, and Surgeon-in-Chief of the Tisch Cancer Hospital at The Mount Sinai Hospital