In this dynamic state, they serve as guardians, clearing away damaged cells, pruning unneeded synapses, and producing molecules that help support neuronal health.
As the brain ages, however, microglia are among the first cells to show signs of decline. Their appearance and function change in ways that can have profound effects on brain health. With age, microglia switch on genes linked to inflammation and immune activation, while the genes that keep them in a stable, balanced state become less active. Their ability to clear waste diminishes, and their energy systems become less efficient.
One particularly important process that falters is autophagy, the cell’s built-in self-cleaning system. When autophagy weakens, microglia begin to accumulate debris, such as damaged mitochondria and protein clumps. Instead of protecting the brain, these overloaded cells may contribute to inflammation and dysfunction. Increasingly, researchers believe that such age-related changes in microglia may not simply reflect brain aging but may actually help drive it.
Recent discoveries from the lab of physician-scientist Anne Schaefer, MD, PhD, Professor of Neuroscience, and Psychiatry, have added a surprising new dimension to our understanding of microglia. Beyond their well-known roles in immune surveillance and cleanup, microglia also help regulate neuronal activity. They do this through the production of a neuromodulatory molecule called adenosine. Microglia generate adenosine (see Figure 1, left panel) by breaking down adenosine triphosphate (ATP) released by neurons and another glia cell, called astrocyte, during activity.
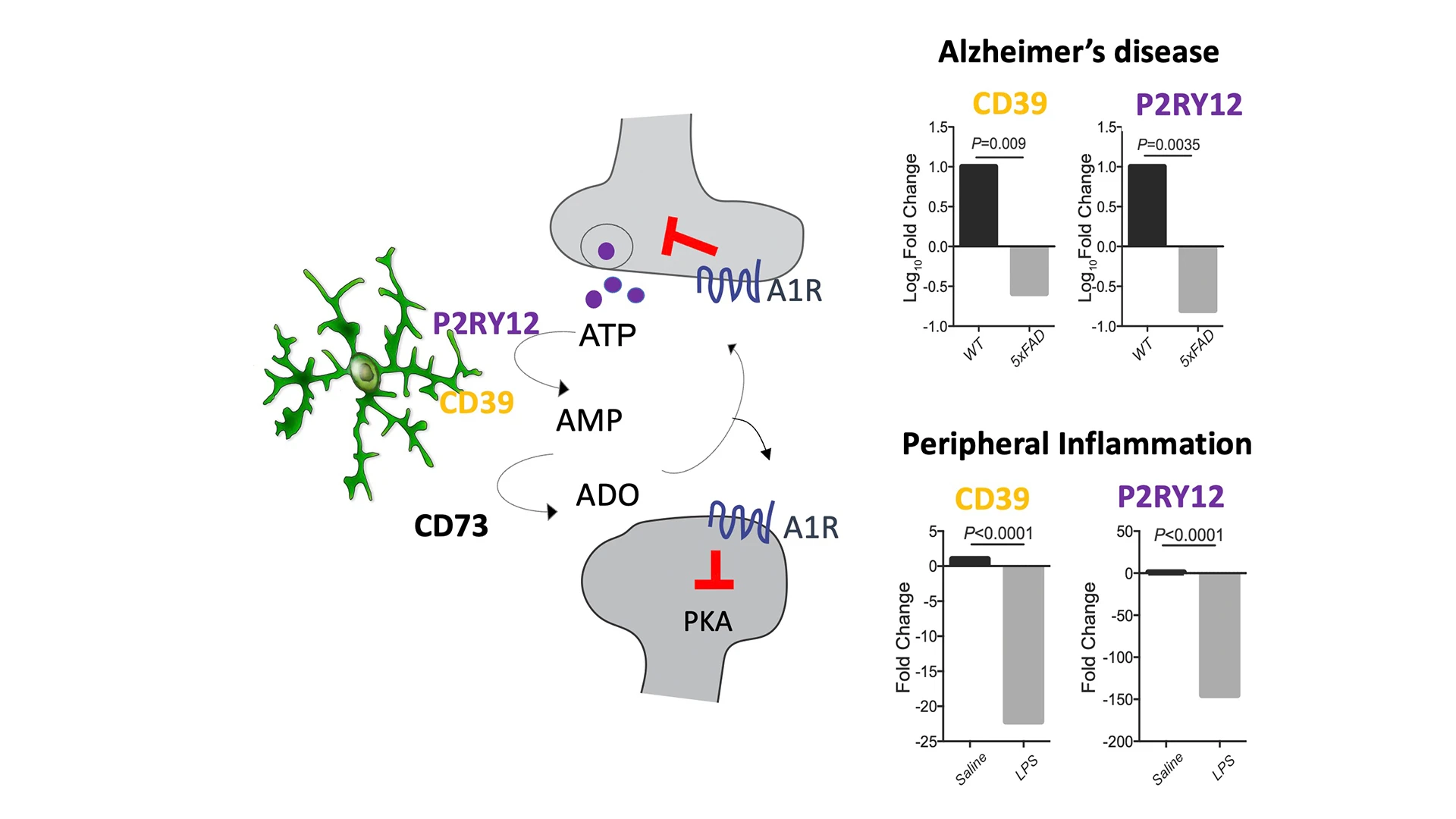
Figure 1. Negative feedback control of neuronal activity by microglia-produced adenosine. Model of activity-dependent microglia-mediated adenosine-controlled regulation of neuronal responses in the healthy brain. Left panel: This shows microglia (green) engaging with neuronal synapses (gray). Right panel: This important neuromodulatory mechanism is downregulated upon microglia activation during inflammatory and/or neurodegenerative diseases.
Under normal conditions, adenosine helps keep neurons from becoming overactive, provides protection during stress, and supports vital processes such as restorative sleep and memory formation. But with age or in disease (see Figure 1, right panel), microglia lose their ability to produce sufficient adenosine. The result is a cascade of problems, including neuronal hyperactivity, sleep cycles that are disrupted, and memory that declines. Recent unpublished findings from the Schaefer Lab open up the intriguing possibility that boosting microglial adenosine signaling could help preserve brain health into old age.
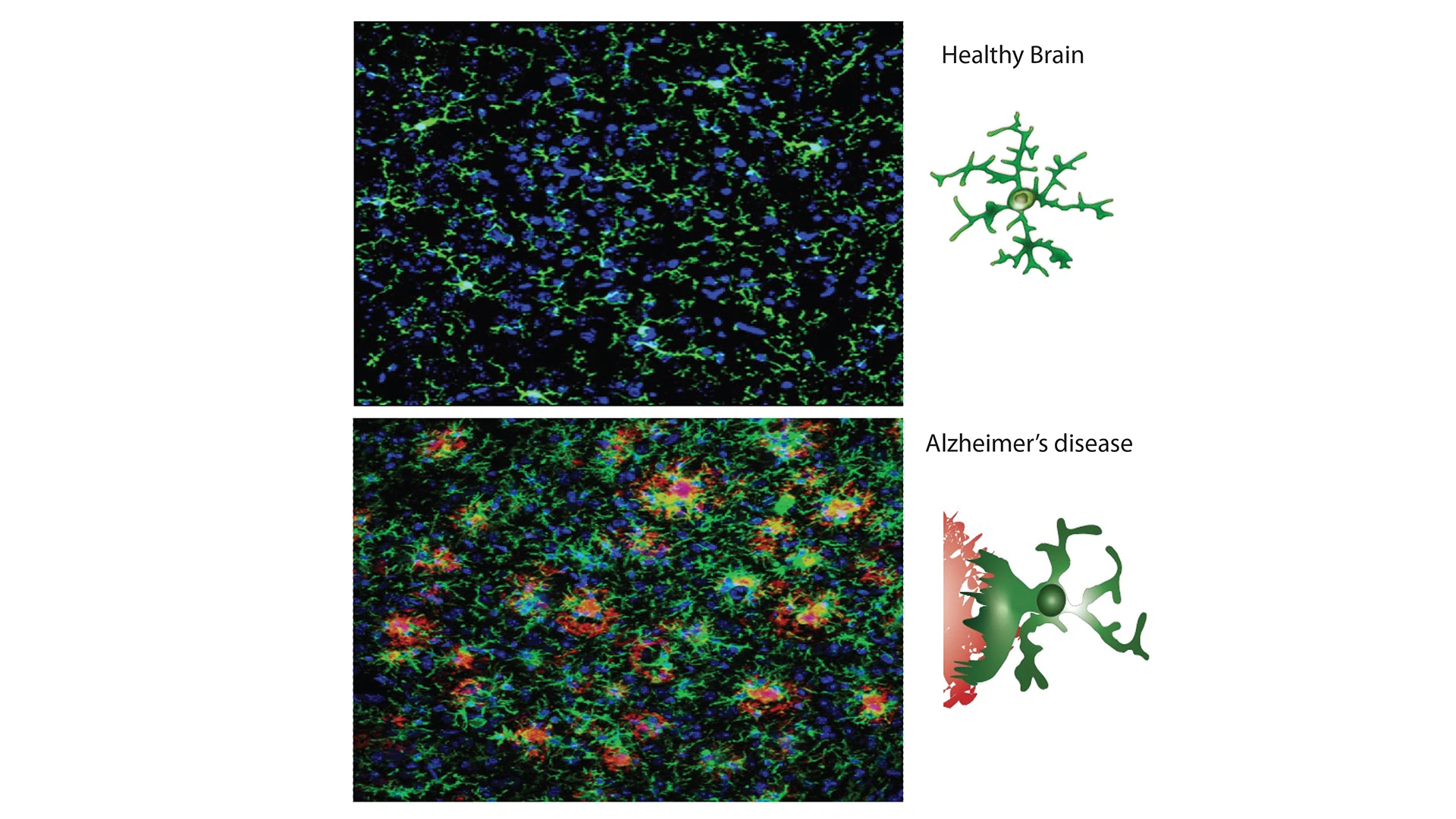
Figure 2. Microglia activation in Alzheimer’s disease (AD). Microglia (green) in the cortex of a healthy brain (top) become activated in the cortex of a mouse model of AD (bottom, blue cell nuclei). Schematic shows microglial activation and morphology changes when responding to beta-amyloid plaques (red).
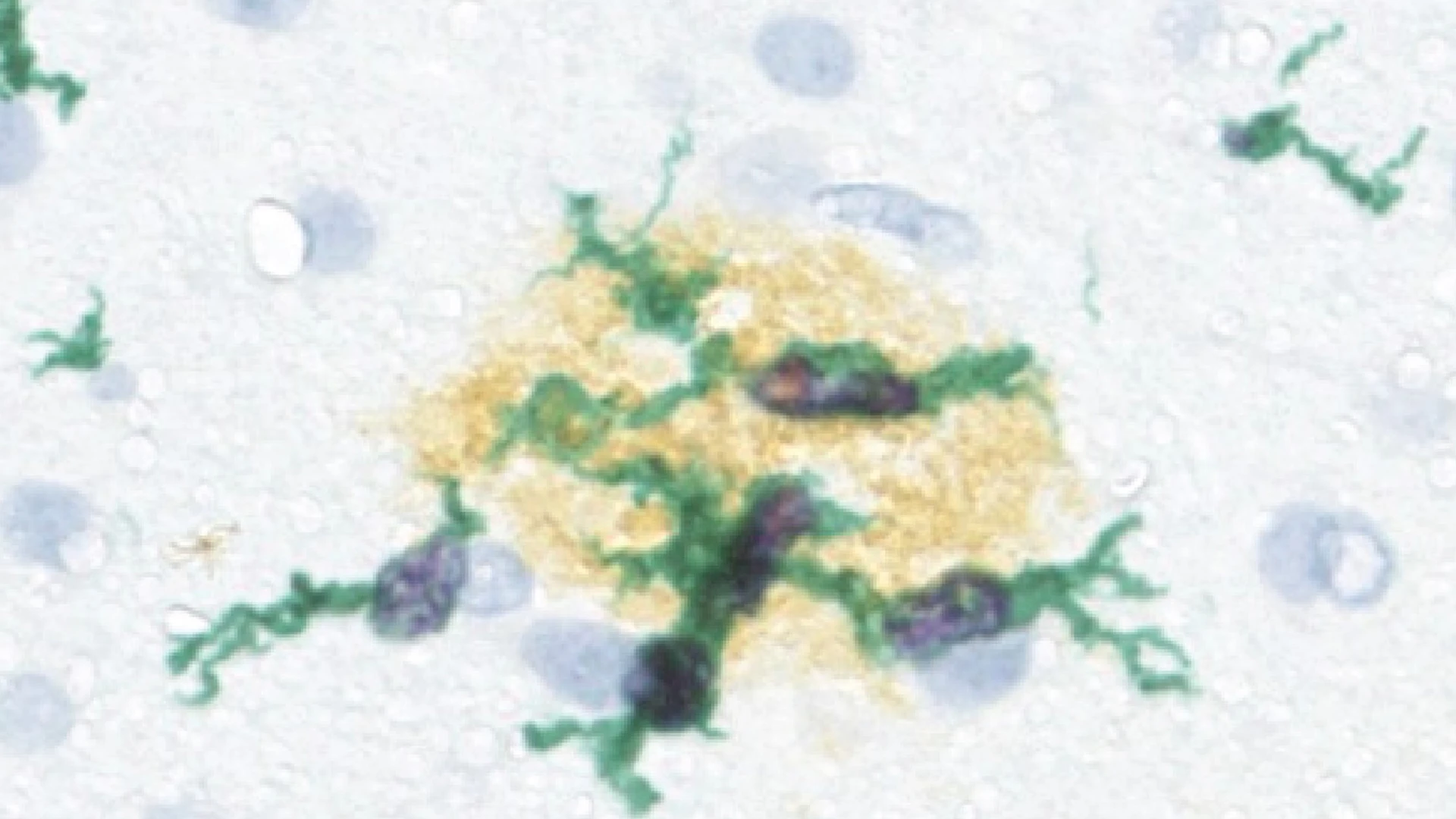
The decline of microglial function is particularly evident in neurodegenerative conditions such as AD (see Figure 2) and Parkinson’s disease (PD). In these conditions, microglia are chronically activated, attempting to clear harmful protein aggregates such as amyloid-β (see Figure 3) and tau in AD and or α-synuclein in PD, while their ability to clear these proteins declines.
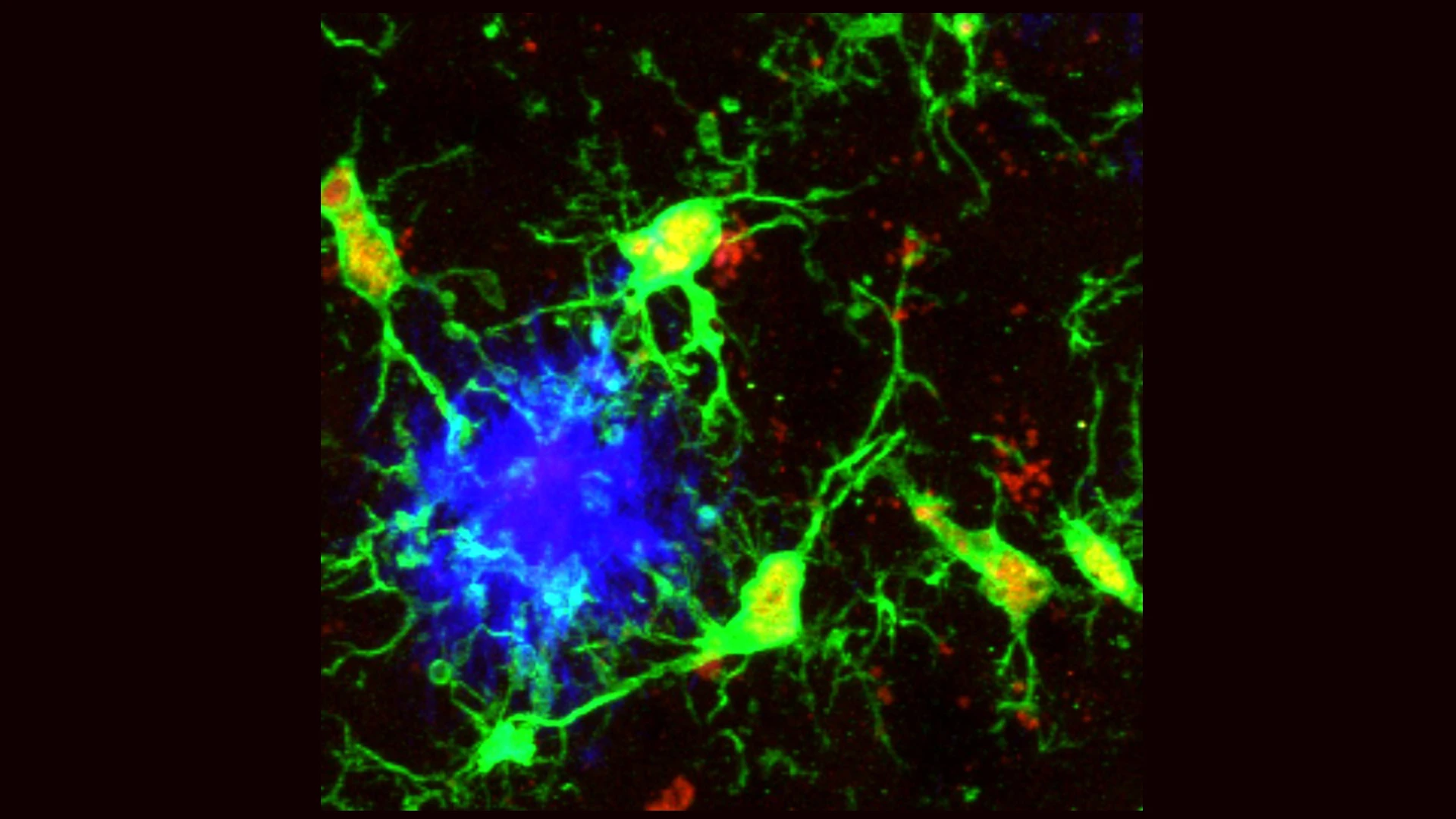
Figure 3 (above and below). Microglia sensing and attacking amyloid plaques in the mouse and human brain. Microglia (green) sensing and responding to aggregated beta-amyloid plaques (blue) in the mouse cortex (above) or amyloid plaques (brown, below) in human diseased brain tissue (blue cell nuclei staining).
Microglia can become locked in a chronic inflammatory state that actually worsens damage, resulting in diminished production of adenosine, deficits in their ability to clear cellular debris, and an increase in their production of inflammatory signals. This shifts microglia from guardian to adversary mode, potentially explaining why patients often experience symptoms such as sleep disturbances, mood changes, and memory problems long before neurodegeneration is formally diagnosed.
Adding further complexity, microglia are not a uniform population of cells. Research by the Schaefer Lab and others has revealed that distinct microglial subtypes exist with specialized roles.
One group, known as disease-associated microglia (DAM), often clusters around amyloid plaques in humans and mouse models of AD. Initially, these cells appear to help, but in chronic disease settings, they lose effectiveness and may even contribute to pathology. Understanding the molecular switches that control whether microglia act in protective or harmful ways has become a central priority for the field. If scientists can identify and manipulate these switches, it may be possible to reprogram microglia, thereby restoring their beneficial functions without undermining their essential role as immune sentinels.
In support of this idea, the Schaefer Lab identified a previously unrecognized protective state of microglia that may open new avenues for treating AD. Using AD mouse models, human cells, and human brain tissue, the team identified a small microglia subpopulation that expresses low levels of the transcriptional regulator PU.1 and induces the expression of immunoregulatory lymphoid receptor proteins, including CD28. Much like suppressor T cells that control autoimmunity in the peripheral immune system, this PU.1-low CD28-expressing microglial population can control brain-wide microglial inflammation and slow the buildup of amyloid plaques. This discovery reveals a new immunoregulatory pathway in the brain that could be harnessed to develop therapies enhancing microglial protective functions to slow or prevent AD progression (see Nature, November 2025).
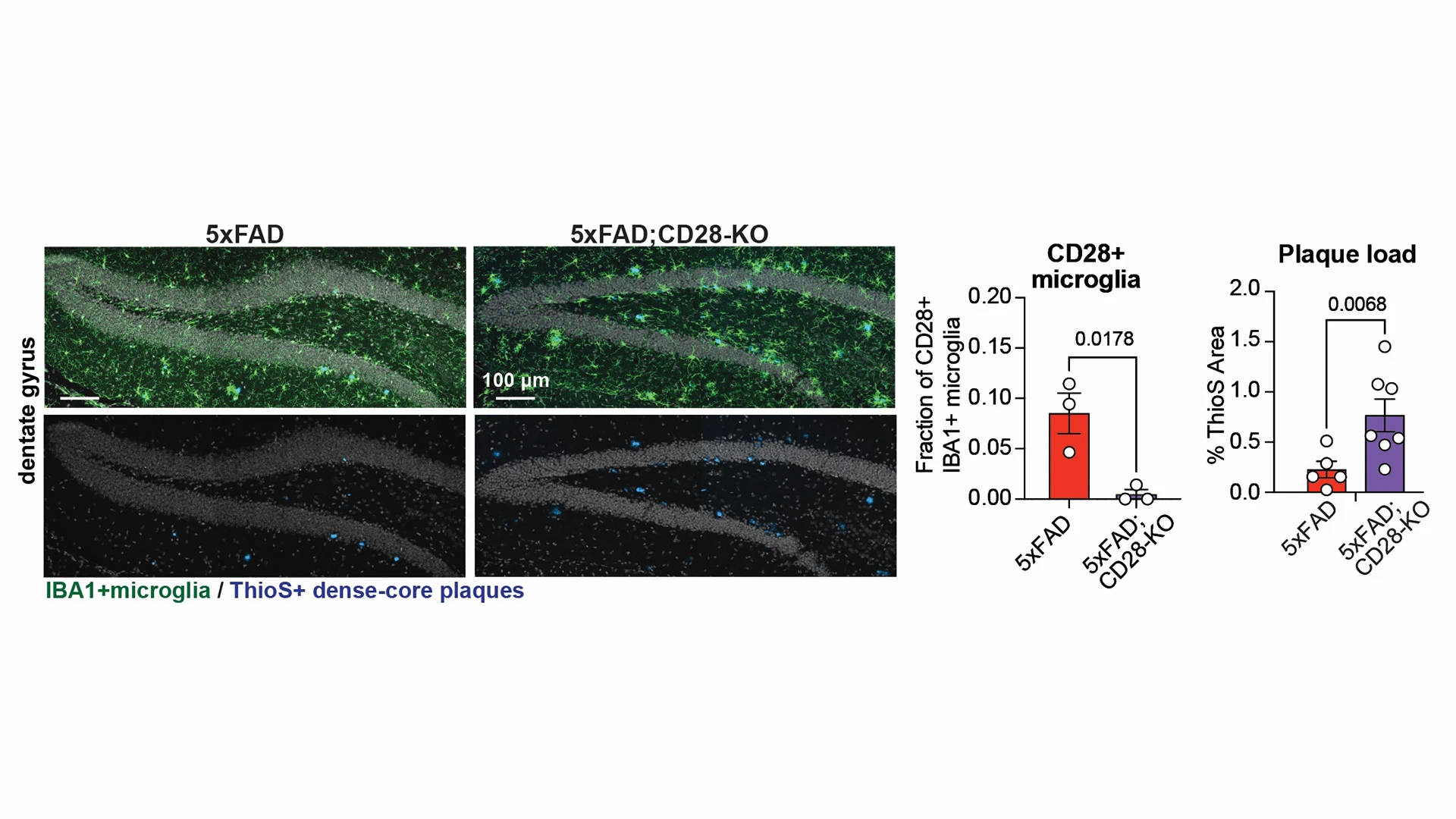
Figure 4. Identification of an immunoregulatory neuroprotective microglia may open new path for immunotheraphy approaches in AD. A small CD28-expressing microglial population controls brain-wide microglial inflammation and slows the buildup of amyloid plaques. Genetic deletion of CD28 from microglia (CD28-KO, right image and purple bar graphs) in a mouse model of the disease (5xFAD) increases microglia (green) inflammatory activity and plaque (blue) formation in mice. Quantifications are shown on the right.
With this goal in mind, researchers at Mount Sinai continue to explore innovative approaches to target microglia.
One strategy involves temporarily depleting existing microglia using CSF-1 receptor blockers. This “reset” aims to replace dysfunctional cells altogether and allows new, healthier microglia with more youthful characteristics to repopulate the brain.
Another approach is microglia transplantation, which aims to introduce neuroprotective microglia with disease regulatory potential.
A third strategy looks at epigenetic reprogramming or targeted drug treatments that could reset aged microglia to a younger, more protective state. Because microglia are highly adaptable, scientists are optimistic that interventions could have meaningful effects even when started later in life.
The fourth strategy is based on the most recent findings that suggest modulation of immunoregulatory receptors on microglia could form the basis of new immunotherapeutic approaches for AD and other neurodegenerative disorders, akin to the checkpoint immunotherapy paradigms revolutionizing cancer treatment.
Yet challenges remain. Microglia must continue to perform their critical functions, even as therapies attempt to correct what has gone wrong. Any potential treatment must be able to reach the brain, act at the right time, and avoid interfering with the immune system’s response to real threats. Reliable biomarkers that track microglial activity will be crucial for guiding and monitoring these new therapies.
The emerging picture is one of hope. Brain aging may not be as inevitable as once believed. Microglia, long overlooked as mere housekeeping cells, are increasingly recognized as powerful regulators of brain health. They can shape neuronal circuit activity and determine whether the brain remains resilient or succumbs to disease. By finding ways to preserve their protective roles while preventing their decline, researchers aim to keep our minds sharper, our sleep deeper, and our brains more resilient. The future of brain health may depend on harnessing the remarkable plasticity of microglia to ensure these guardians remain allies, not adversaries, in the aging brain.
Featured

Anne Schaefer, MD, PhD
Professor of Neuroscience, and Psychiatry, Icahn School of Medicine at Mount Sinai, and Co-Director, Center for Glial Biology, The Friedman Brain Institute
