Neuroscientists have probed the cellular and molecular foundations of this complex neural process for decades. Recent discoveries challenge two long-standing assumptions: first, the notion that memory loss and brain aging are immutable processes, and second, there is limited body-brain communication shaping these processes. However, exciting advances from the lab of Joseph M. Castellano, PhD, at The Friedman Brain Institute highlight youth-associated blood proteins that restore brain function in aged animals.
A seminal discovery at the heart of these findings took advantage of a classical surgical technique known as parabiosis. The studies involved joining aged and young animals, allowing their blood circulation to be shared for many weeks. Exposing an aged animal to young circulatory factors in this way was sufficient to improve neural function, including the birth of new neurons and the strengthening of synapses, in the form of long-term potentiation and increased dendritic spine number. Importantly, these changes were seen in the hippocampus, a key brain region involved in learning and memory processes.
Exciting advances from the Castellano Laboratory at The Friedman Brain Institute highlight youth-associated blood proteins that restore brain function in aged animals.
These findings were notably expanded in experiments involving the transfer of plasma, the soluble component of blood, which significantly improved not only synaptic plasticity, but also memory performance. Treated mice that were 18 months old, an age roughly corresponding to 65-year-old humans, were able to navigate several tasks for hippocampus-dependent memory in a manner that resembled younger mice. Notably, heat denaturation of proteins within plasma appeared to eliminate these memory improvements, igniting a search for so-called “rejuvenating” factors that are sufficient to provide the beneficial effects of young plasma.
When researchers modeled the TIMP2 deficiency seen with age, the generation of newborn neurons in the adult hippocampus was significantly reduced, the mice had less complex dendrites, and they were less able to perform various memory tasks...
To filter the enormous list of possible factors, researchers, led by Dr. Castellano, Associate Professor of Neuroscience, and Neurology, searched for proteins changing across lifespan in both humans and in animal models to identify conserved proteins elevated in plasma in very early life (see Nature, 2017). This search turned up a surprising candidate called tissue inhibitor of metalloproteinases 2, or TIMP2, which they found enters the brain from blood. Supplementing aged mice with this protein systemically to mimic the plasma transfer paradigm produced remarkably similar results—improved synaptic plasticity and memory performance.
Aside from a handful of studies describing its high hippocampal expression and possible roles in motor coordination, little was known about how this protein acts in the brain and how its activity restores function in the context of aging.
One clue came from its role in development. Its expression is highly upregulated in various tissues during early-life developmental stages. Exposing aged animals to youth-associated factors such as TIMP2 may rekindle developmental processes in neural cells that become quiescent later in life. Indeed, when researchers in the Castellano Laboratory modeled the TIMP2 deficiency seen with age, the generation of newborn neurons in the adult hippocampus was significantly reduced, the mice had less complex dendrites, and they were less able to perform various memory tasks that depend on adult neurogenesis, including spatial memory discrimination tasks (see Molecular Psychiatry, 2023). During a complex memory task called the Barnes maze, mice lacking TIMP2 tended to use search strategies that were less reliant on hippocampal function compared to healthy mice.
To understand how TIMP2 mediates such changes, the researchers explored its high-affinity interactions with MMP2, which facilitate conversion of pro-MMP2 to active MMP2, an enzyme that degrades the molecular scaffolding between and around neural cells known as the extracellular matrix (ECM).
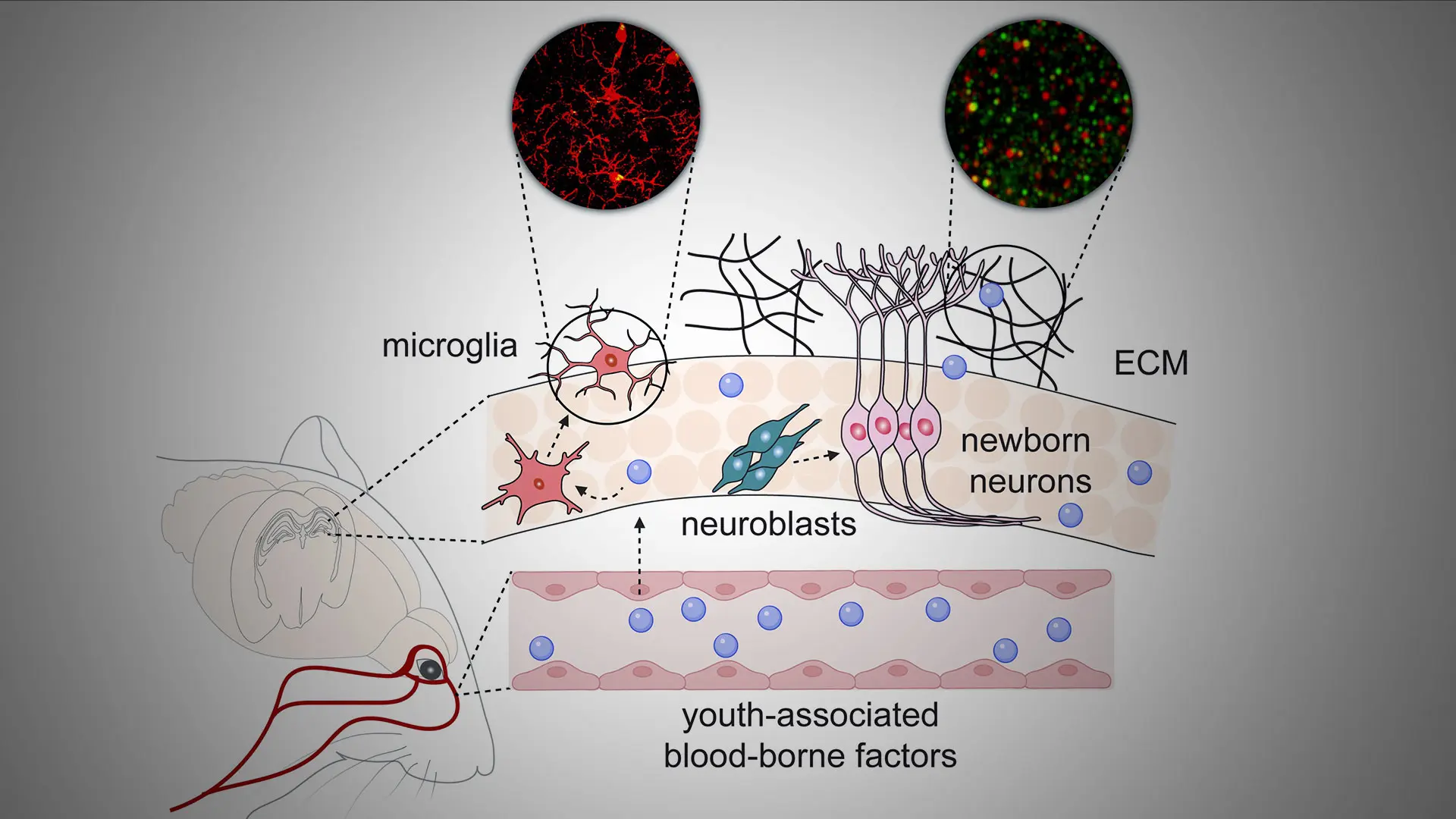
The researchers turned to super-resolution microscopy approaches to visualize the ECM around neurons, finding significant accumulation within TIMP2-deficient hippocampus that mimicked age-related ECM increases. Injecting an enzyme that cleaves side chains of specific ECM components into TIMP2-deficient hippocampus led to a full rescue of the stalled migration of adult neuroblasts. Thus, TIMP2 affects plasticity by interacting with the ECM to maintain its turnover, providing a flexible environment in which neural processes take place that ultimately facilitate memory (see Figure).
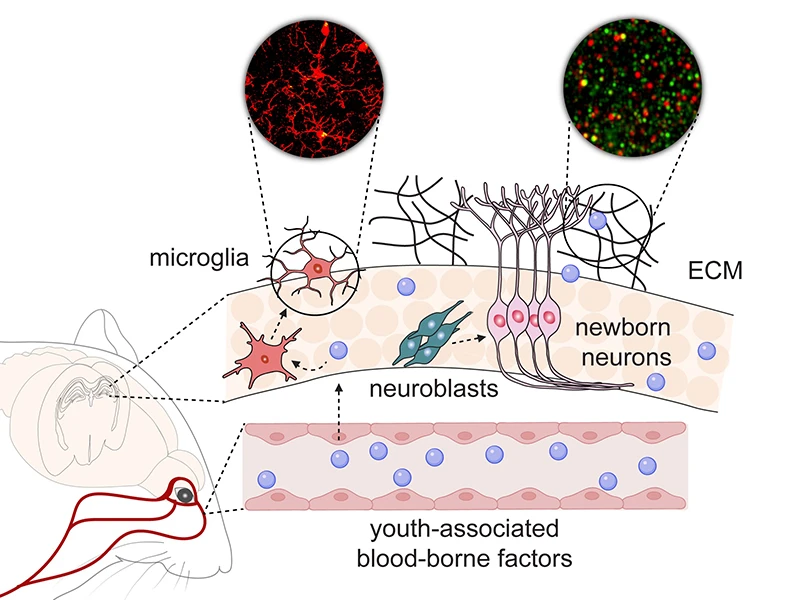
Figure. Youth-associated blood-borne proteins, including TIMP2, act on diverse cellular and molecular targets within the hippocampus to support synaptic integrity, adult neurogenesis, and microglial function.
Other cells of the hippocampal niche are also affected by TIMP2’s activity, including microglia, the brain’s innate immune cells that can adopt dysfunctional states as brains age or exhibit neurodegenerative pathology.
The lab recently showed that losing TIMP2 alters activation states of microglia (see BioRxiv, 2025), while reducing their ability to remove age-associated debris. Providing aged mice with supplementary levels of plasma TIMP2 decreases subsets of microglia expressing markers for dysfunctional states, and the microglia appear to phagocytose material more efficiently. Together, these data argue that youth-associated proteins such as TIMP2 may be viable therapeutic targets for reactivating early-life processes in the aged brain, supporting improved memory and neural function.
With the existing challenge of designing small molecules to cross the blood-brain barrier to reach targets, there is great promise in exploiting such proteins. New work will continue to leverage expansive plasma proteomics to establish relationships between these factors and aging rates of various organs, and personalized approaches should be developed to isolate which factors are the most rejuvenating—and for which diseases—to maximize brain health.
Featured

Joseph M. Castellano, PhD
Associate Professor of Neuroscience, and Neurology, Icahn School of Medicine at Mount Sinai