These discoveries transformed the field, allowing researchers to create cell (see Figure 1) and animal models carrying these mutations, enabling them to gain a better understanding of AD. These endeavors paved the way for the first approved use from the U.S. Food and Drug Administration for amyloid-based immunotherapies for AD.
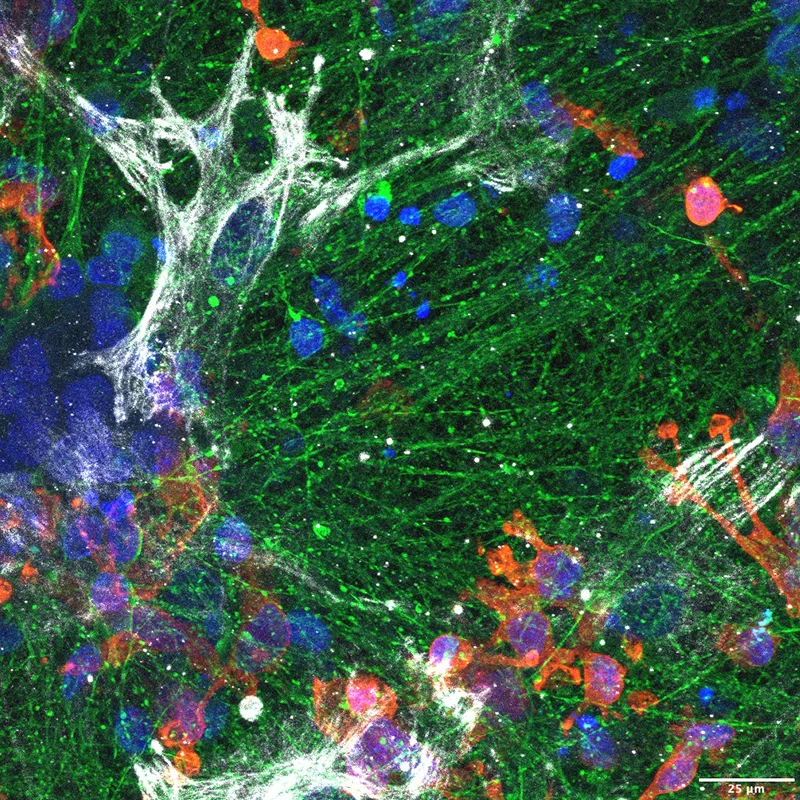
Figure 1. iPSC-derived triculture system. Human induced pluripotent stem cells (iPSCs) are differentiated into neurons (TUJ1, green), astrocytes (GFAP, red), and microglia (IBA1, gray) and cultured together to replicate the multicellular environment of the human brain. This system allows co-culture of astrocytes and microglia with neurons carrying familial AD mutations and their isogenic gene-corrected controls. Credit: Hyo Lee, PhD, and Francesca Garretti, PhD
Apolipoprotein E (APOE) has also been identified as the major genetic risk factor for both early- and late-onset sporadic forms of AD. APOE exists in human populations in three common forms: APOE2, APOE3, and APOE4. APOE3 is the most common isoform in all populations. APOE4 increases risk for AD and reduces age of disease onset while APOE2 decreases risk and increases age of onset. APOE2 was the first protective genotype against AD to be identified.
More recent studies have demonstrated that the magnitude of the effects of APOE4 and APOE2 on AD risk is influenced both by sex and population ancestry. Women are at greater risk compared to men with the same APOE genotype, while Asian populations are at higher risk than Europeans, who in turn are at higher risk than Africans or Hispanics, with the same APOE4 genotype. Like APP/PSENs, these APOE isoforms seem to impact the dynamics of amyloid aggregation and clearance in the brain. However, clinical trials using amyloid immunotherapies have shown that individuals carrying APOE4 benefit less from amyloid-based immunotherapies and exhibit an increased risk of potentially life- threatening side effects, limiting their use in those who are most at risk for AD.
Following decades of research focused on amyloid deposits in the brain as a central driver of AD, recent large-scale genomic studies have uncovered entirely new pathways, opening two exciting new directions for next-generation therapeutics.
First, by comparing the genomes of hundreds of thousands of individuals with AD to those of elderly, cognitively healthy controls, researchers have identified nearly 100 genes that influence disease risk. Strikingly, most of these genes are preferentially expressed in microglia, the brain’s resident immune cells.
Pioneering work from the Alison M. Goate, DPhil, Laboratory has shown that many of these microglia-associated genes regulate the uptake and clearance of lipid-rich cellular debris, a process known as efferocytosis. Impairments in efferocytosis appear to contribute to brain aging and AD pathogenesis. Intriguingly, APOE also plays a critical role in efferocytosis, raising the possibility that dysfunction in this process is a central factor underlying vulnerability to AD. Building on this insight, new therapeutic strategies are being developed to enhance efferocytosis and thereby promote the clearance of cellular debris, including amyloid, from the brain, with the goal of slowing or preventing neurodegeneration.
Following decades of research focused on amyloid deposits in the brain as a central driver of AD, recent large-scale genomic studies have uncovered entirely new pathways, opening two exciting new directions for next-generation therapeutics.
Dr. Goate is the Jean C. and James W. Crystal Professor of Genomics, Chair of the Department of Genetics and Genomic Sciences, and Professor of Neuroscience, and Neurology, at the Icahn School of Medicine at Mount Sinai.
The second promising line of research focuses on DNA variants that naturally protect against AD. This strategy takes advantage of “experiments of nature” to reveal biological mechanisms that safely lower disease risk. Large-scale sequencing studies have already uncovered several rare versions of the APOE gene that, like APOE2, reduce the likelihood of developing AD.
One especially striking case involved a woman who carried a harmful PSEN1 mutation, which typically causes dementia in the early 40s. Remarkably, because she also carried a protective variant called APOE Christchurch, she showed only mild symptoms of dementia when she passed away at age 72. Although this is just one individual, understanding how such variants shield the brain may point to powerful new therapies, including gene-based treatments.
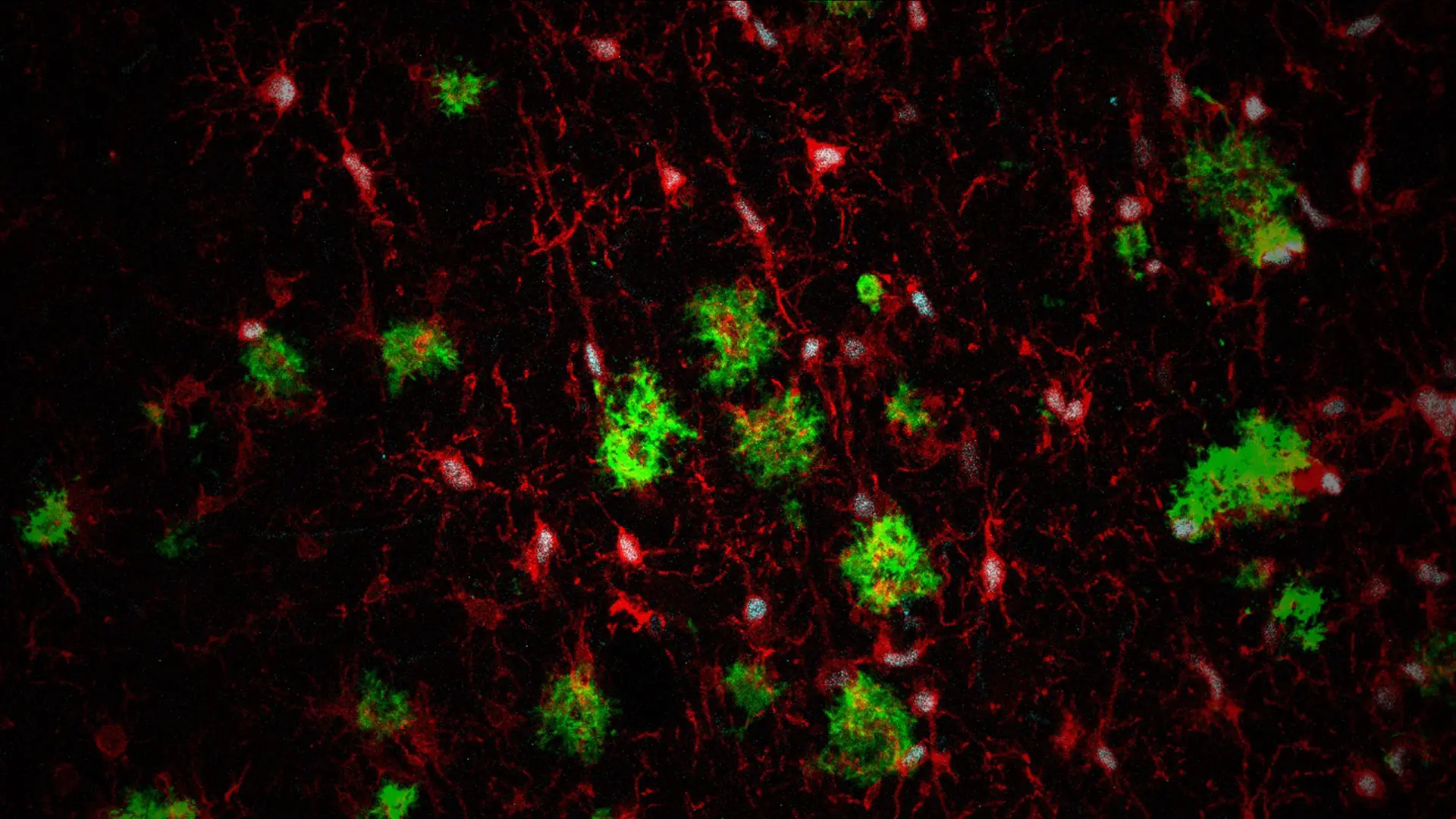
Other strategies include designing antibodies or small molecules that mimic the protective effects of these genetic variants. Leveraging these exciting discoveries, Dr. Goate, along with Yizhou Dong, PhD, Mount Sinai Endowed Professor in Nanomedicine, and Edoardo Marcora, PhD, Professor of Genetics and Genomic Sciences, and Neuroscience, were awarded National Institutes of Health funding to test whether brain delivery of lipid nanoparticles that express a protective APOE variant or suppress risk-associated APOE4 can reduce AD pathology in a mouse model (see Figure 2).
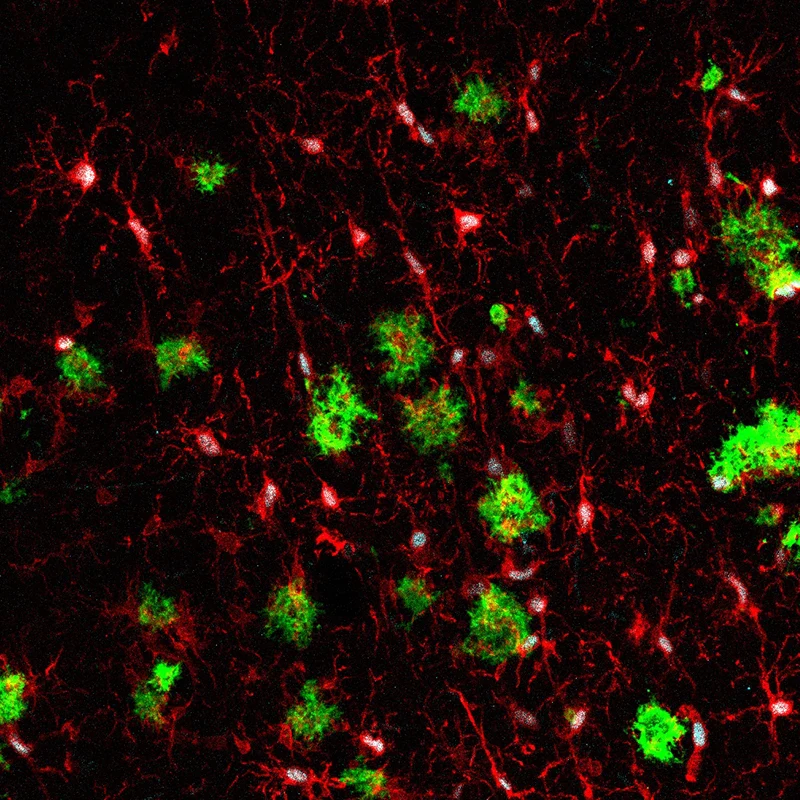
Figure 2. Immunostaining of the brain of a mouse model of AD xenotransplanted with human microglia. Green: amyloid plaques; red: microglial cells; cyan: marker of human microglia. Credit: Carmen Romero-Molina, PhD
With such breakthroughs in AD genetics, the coming decades hold extraordinary promise for developing new therapies that target previously untapped disease mechanisms to protect against—and even reverse—AD pathology.
Featured

Alison M. Goate, DPhil
Jean C. and James W. Crystal Professor of Genomics, Chair of the Department of Genetics and Genomic Sciences, and Professor of Neuroscience, and Neurology, Icahn School of Medicine at Mount Sinai