Others (knowing of The Monkey’s Paw by W.W. Jacobs or its inspirations) might pessimistically foresee a bargain where they eventually become burdened with multiple neurodegenerative, motor, and other diseases for eternity.
As improvements in public health and medical science have afforded dramatic increases in life expectancy within the past century, many are already contending with a version of this scenario, and most would prefer to live as long a life as possible provided they maintain intact cognition and the capacity to enjoy that long life.
An ongoing controversy is whether aging itself causes cognitive decline, or whether almost all age-associated changes stem from disease pathologies that grow more likely with time. At the Icahn School of Medicine at Mount Sinai, several studies use advanced neuroimaging to disentangle these questions and uncover strategies to preserve cognitive health.
The Alzheimer’s Disease Research Center (ADRC) at Mount Sinai, funded by the National Institute on Aging, has examined people at risk for Alzheimer’s disease (AD) for more than 40 years, following some individuals across decades. Until recently, AD pathology could only be confirmed at autopsy. Since the 2010s, positron emission tomography (PET) tracers for amyloid plaques and tau tangles have allowed researchers to track these pathologies in the living brain. Coupled with magnetic resonance imaging (MRI) and additional PET tracers, these methods now provide richly detailed brain characterization of brain aging.
In the laboratory of Trey Hedden, PhD, Core Lead of the Neuroimaging Biomarker Core of the ADRC, nearly 200 older individuals have been scanned with combinations of amyloid PET, tau PET, dopamine PET, fluorodeoxyglucose (FDG) PET, and multiple MRIs, making them among the most comprehensively studied brains worldwide. Dr. Hedden refers to these volunteers as “brain-STARS” (Studied for Tau, Amyloid, Receptors, and/or Synapses). Such intensive characterization is enabled by a cohort of enthusiastic research participants and by the PET-MR scanner at the Biomedical Engineering and Imaging Institute at Mount Sinai, an instrument installed at a relatively small number of institutions.
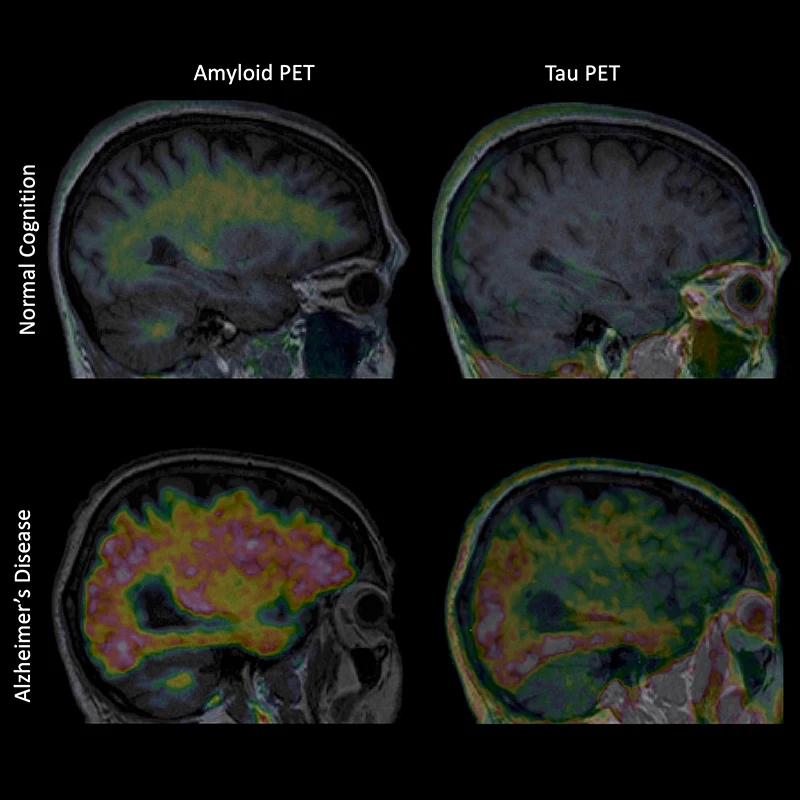
Figure 1. Amyloid and tau PET images from an older individual with normal cognition, and from a patient with AD.
Research now shows that much of what has been considered to be age-related memory loss may actually reflect undiagnosed AD. Individuals with amyloid plaques exhibit memory test declines years before diagnosis of mild cognitive impairment. Those with both amyloid and tau exhibit even more rapid memory decline. Recently developed blood tests can detect tau fragments, offering a less invasive way to indicate AD pathology. In collaboration with physician-scientist Fanny Elahi, MD, PhD, Dr. Hedden is evaluating how such blood assays can guide decisions about whether PET imaging should be performed. As these tools become more available, AD may be detected at its onset—and perhaps treated before memory decline appears.
Anti-amyloid therapies can remove plaques, though their benefit for memory preservation remains modest and risks can be significant. Tau-targeting therapies are also in development, raising hope that early detection combined with intervention may one day prevent memory loss from AD altogether.
Genetics adds another critical dimension. In collaboration with the Ronald M. Loeb Center for Alzheimer’s Disease and the Sanford Grossman Interdisciplinary Center in Neural Circuitry and Immune Function, Dr. Hedden’s studies incorporate genetic profiling. The APOE-e4 allele remains the strongest single risk factor for AD, but polygenic risk scores summing contributions across many genes are increasingly informative. Early findings suggest that individuals with high polygenic risk but no APOE-e4 allele may still show elevated amyloid on PET, underscoring how AD can arise from multiple genetic vulnerabilities.
Other age-related brain changes also weigh on cognition. Using a PET tracer, Dr. Hedden measures dopamine receptor density in the caudate and putamen, key regions for motor function and learning. Dopamine declines during aging and is profoundly disrupted in Parkinson’s disease (PD) and Lewy body dementia (LBD). These dopamine changes, linked to memory and executive function, may arise from undetected alpha-synuclein pathology long before clinical symptoms. Dr. Hedden is pursuing funding to determine whether such dopaminergic declines are related to alpha-synuclein in individuals without PD or LBD.
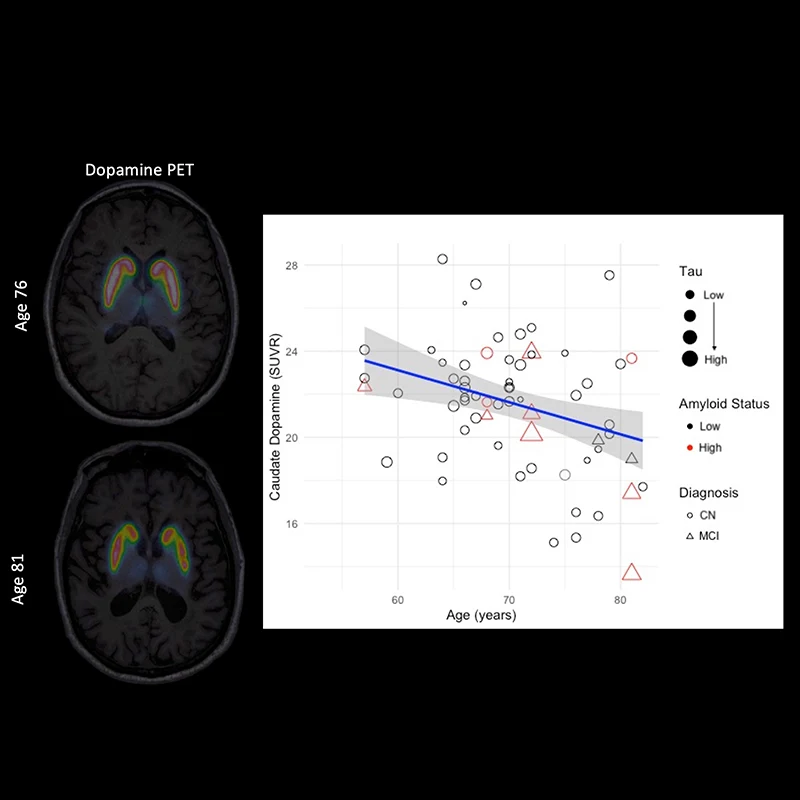
Figure 2. Dopamine PET images showing D2/D3 receptor binding in caudate and putamen. Graph illustrates age-related decreases in receptor binding, distinct from amyloid, tau, or diagnosis.
Cerebrovascular health is equally critical. White matter lesions, often linked to hypertension, disrupt communication and impair memory, executive function, and processing speed. Recent work by Dr. Hedden and collaborator Xiang Xu, PhD, found that cerebral blood flow is impaired in regions with elevated amyloid, suggesting vascular disease and amyloid pathology may interact, possibly via amyloid deposits in blood vessel walls.
Taken together, Dr. Hedden’s research shows that these factors—amyloid, tau, dopamine signaling, vascular health, and genetics—interact to determine cognitive trajectories. Each alone may impair cognition, but in combination they accelerate decline.
By improving early detection with imaging and biomarker tests, researchers aim to intervene earlier, preserve cognition, and maximize the chance of maintaining brain health for a lifetime.
Featured

Trey Hedden, PhD
Associate Professor of Neurology, Neuroscience, and Diagnostic, Molecular and Interventional Radiology; and Director of Neuroimaging and Biomarker Research in Aging and Alzheimer's Disease