Mount Sinai researchers have identified a cellular mechanism linking infections from influenza A viruses to cardiovascular disease. The findings provide critical insights on how influenza can damage the heart and increase the risk of a heart attack or other major cardiovascular event, says Filip K. Swirski, PhD, Director of the Cardiovascular Research Institute at the Icahn School of Medicine at Mount Sinai and the senior author of the study.
Through its work with mouse models and human data, the team also provided evidence that a leading-edge modified mRNA treatment that dampens an interferon signaling pathway in the heart can significantly mitigate cardiac damage following viral infection while preserving the protective antiviral response of the immune system. The study was published in February 2026 in the journal Immunity.
“We have known for years that the frequency of heart attacks increases during flu season, yet outside of clinical intuition, scant evidence exists of the underlying mechanisms of that phenomenon,” Dr. Swirski says. “Studies like ours are now shedding valuable light on immune system pathways, like the antiviral cytokine type 1 interferon (IFN-1), that factor into damage to the heart following severe influenza infection. These findings offer great promise for the development of new therapies, which are desperately needed since there are currently no viable clinical options to prevent cardiac damage.”
The Mount Sinai team studied autopsies of 35 hospitalized patients who died of influenza and found that more than 85 percent had at least one significant cardiovascular comorbidity, such as hypertension. They also found that the majority had multiple comorbidities, including atherosclerosis and cardiac fibrosis, underscoring cardiovascular disease as a major driver of influenza mortality.
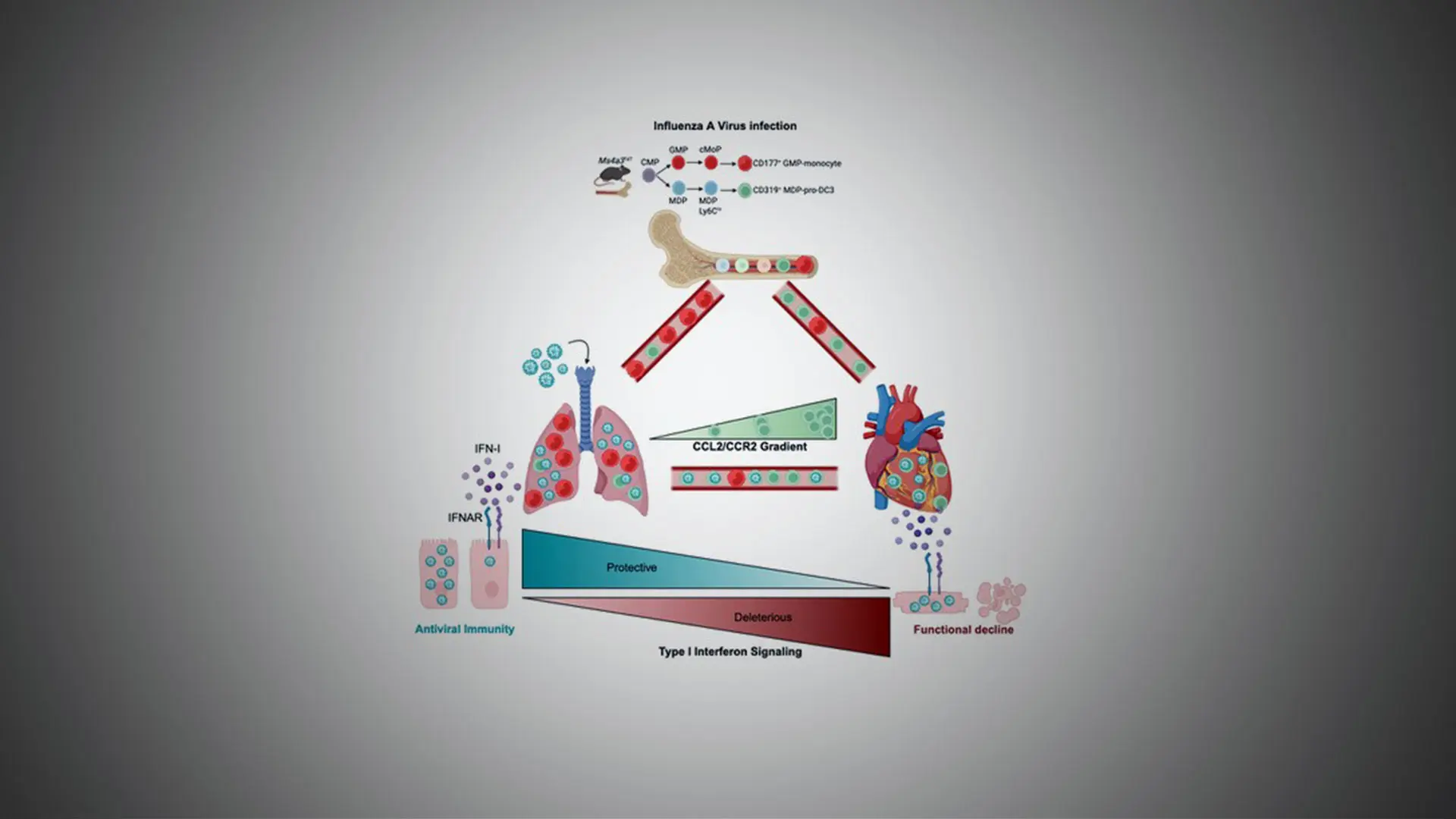
The research team also uncovered the mechanism by which cardiac damage occurs. They learned, for example, that a novel subset of white blood cells, known as pro-dendritic cell 3 (pro-DC3), becomes infected in the lung and, after traveling to the heart, produces large amounts of type 1 interferon. This, instead of fulfilling its mission of clearing the virus from the heart, triggers the death of cardiomyocytes, impairing cardiac output.
“Better understanding of influenza pathogenesis and immune pathways that are activated throughout the body will help fuel the next stage of advanced care.”
Filip K. Swirski, PhD
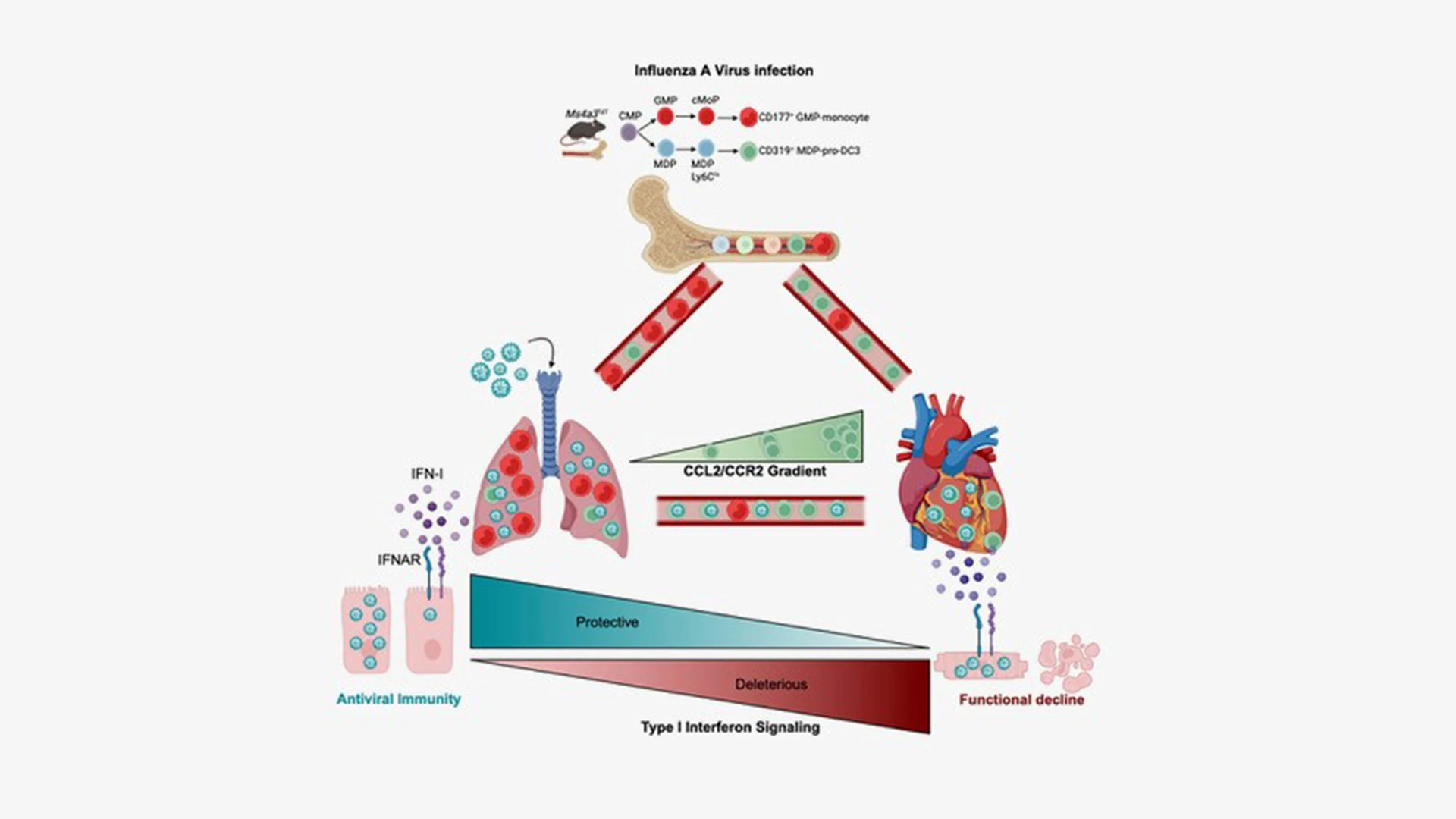
The authors found that severe influenza damages the heart by exploiting specific immune cells and engaging a type I interferon response. The authors also show that therapeutic silencing of the response mitigates heart damage.
“We found that the pro-dendritic cell 3 acts as the ‘Trojan horse’ of the immune system during influenza infection, becoming infected in the lung, trafficking the virus to the heart, and disseminating it to cardiomyocytes. This process causes production of the damaging type 1 interferon that comes with considerable collateral damage to the heart,” explains Jeffrey Downey, PhD, a member of Dr. Swirski’s laboratory who served as lead author of the study. “The hopeful news for patients is that by injecting a novel mod-RNA therapeutic that modulates the IFN-1 signaling pathway, we reduced levels of cardiac damage, as evidenced by lower troponin, and improved cardiac function, as measured by higher left ventricular ejection fraction.”
As part of its ongoing research, Dr. Swirski’s team is collaborating with Lior Zangi, PhD, Associate Professor of Medicine (Cardiology), and Genetics and Genomic Sciences at the Icahn School of Medicine at Mount Sinai, to investigate the use of a safe and effective systemic delivery method of the mod-RNA therapeutic to the heart’s muscle cells, instead of the direct injection method used in its proof-of-concept study. Additional work is focused on the pro-dendritic cell 3 itself: why is it so susceptible to influenza and how could its protective capacity be fully harnessed to potentially minimize heart damage exacerbated by cardiovascular disease?
“As IFN-I is broadly antiviral, our study’s insights may extend far beyond influenza. Moreover, we have shown that vaccination, even if only partially protective in preventing overt morbidity with a mismatched virus, protects from heart damage, thus extending the benefits of vaccination outside of the well-documented protection of the lung,” the study concluded. “In sum, we have shown that influenza hijacks a specific monocyte-like pro-DC3, seeds the heart, infects cardiomyocytes, and induces IFN-I production that damages the heart. Harnessing this pathway has the potential to minimize viral heart damage exacerbated by cardiovascular disease.”
“Pathogens are constantly emerging and evolving, which means our strategies to combat them must evolve, as well,” Dr. Swirski says. “Better understanding of influenza pathogenesis and immune pathways that are activated throughout the body will help fuel the next stage of advanced care.”
This study was principally supported by the following grants from the National Heart, Lung, and Blood Institute and the Charles H. Revson Foundation: P01HL131478, P01 HL142494, R01 HL178835, K99 HL177314, and 24-29.
Featured

Filip K. Swirski, PhD
Director of the Cardiovascular Research Institute, Arthur and Janet C. Ross Professor of Medicine (Cardiology), and Professor of Diagnostic, Molecular and Interventional Radiology

Jeffrey Downey, PhD
Swirski Laboratory Fellow

Lior Zangi, PhD
Associate Professor of Medicine (Cardiology), and Genetics and Genomic Sciences