The Cardiovascular Research Institute (CVRI) in 2025 continued to advance the understanding of the heart and blood in a broad physiological context—including new insights into repair after injury—under the leadership of Director Filip K. Swirski, PhD.
“Our teams are taking a highly interdisciplinary, whole-body approach to their research because we know that heart disease results from an interaction among several physiological systems,” says Dr. Swirski, the Arthur and Janet C. Ross Professor of Medicine (Cardiology), Diagnostic, Molecular and Interventional Radiology, and Immunology and Immunotherapy at the Icahn School of Medicine at Mount Sinai. “Understanding the mechanisms of inter-system communication is key to innovation in fundamental and translational science.”
Among significant achievements, Hina Chaudhry, MD, and team found in a breakthrough study that that the gene Cyclin A2 (CCNA2), which turns off after birth in humans, can make new, functioning heart cells and help the heart repair itself from injury, including a heart attack or heart failure, when the gene is turned back on. And a team led by Lior Zangi, PhD, used a targeted, nonviral modified mRNA system to transiently boost expression of a regenerative enzyme, pyruvate kinase muscle isozyme M2 (PKM2), in heart muscle cells of juvenile and adult pig models after ischemic injury.
David Goukassian, MD, PhD, and team continued their study of the effects of space irradiation on the heart during space missions, finding long-lasting and sex-specific alteration in left ventricular cardiac transcriptome. And a team led by Cameron McAlpine, PhD, used mouse models and a human trial to test if hematopoiesis, the process by which immune cells are produced, links cardiometabolic syndrome and sleep with multiple sclerosis and neuroinflammation.
The investigators provide insights into their work below:
Abstract of study:
Transient overexpression of hPKM2 in porcine cardiomyocytes prevents heart failure after myocardial infarction. Nature Communications, November 2025.
The adult mammalian heart lacks the ability to regenerate after injury, contributing to heart failure. No current treatment reactivates heart muscle cell division to prevent this decline. We used a targeted, non-viral modified mRNA system to transiently boost expression of a regenerative enzyme, pyruvate kinase muscle isozyme M2, in heart muscle cells of juvenile and adult pig models after ischemic injury. In juvenile pigs treated one-week post-injury, we observed increased markers of cell division, secretion of protective factors, improved heart function, and reduced scarring two months later. In adult pigs treated immediately after injury, we saw improved heart contractility and less fibrosis one month later. These results show that targeted pyruvate kinase muscle isozyme M2 modified mRNA delivery can stimulate muscle regeneration and functional recovery in both young and adult pig hearts. This approach offers a promising strategy for repairing ischemic injury and preventing heart failure in humans.
Notes From the Investigator: Lior Zangi, PhD
This study shows that hPKM2 modRNA can drive cardiomyocyte proliferation, reduce scar formation, and preserve cardiac function after ischemic injury. By combining the immune-evasive properties of modRNA with our cardiomyocyte-specific delivery platform, we achieved selective, transient hPKM2 expression in cardiomyocytes. This platform enables regenerative, protective, and angiogenic signaling while minimizing off-target effects.
Our success in both juvenile and adult pig models is particularly important. Age remains one of the largest barriers to tissue regeneration, and most regenerative strategies lose efficacy as tissues mature. Demonstrating robust functional preservation in adult pigs, whose cardiac structure, physiology, and post-infarct remodeling closely resemble humans, shows that the therapy remains effective even in mature hearts, where regenerative capacity is especially limited.
From a mechanistic standpoint, this work highlights PKM2 as more than a proliferative trigger. Our studies show that PKM2 integrates metabolic rewiring, redox control, and cell-cycle activation in cardiomyocytes, allowing controlled and transient regeneration. The cardiomyocyte-specific platform is central to this effect. Without targeted and transient delivery, activation of this pathway would carry significant risk. The data show that our treatment induces regeneration while preserving myocardial architecture and function.
The next steps are focused on moving toward clinical application. This includes further optimization of the hPKM2 modRNA payload, refinement of dosing, and continued optimization of delivery methods to support scalability and safety. Together, these efforts are aimed at advancing this platform toward human clinical testing and enabling a new approach to myocardial repair after ischemic injury.
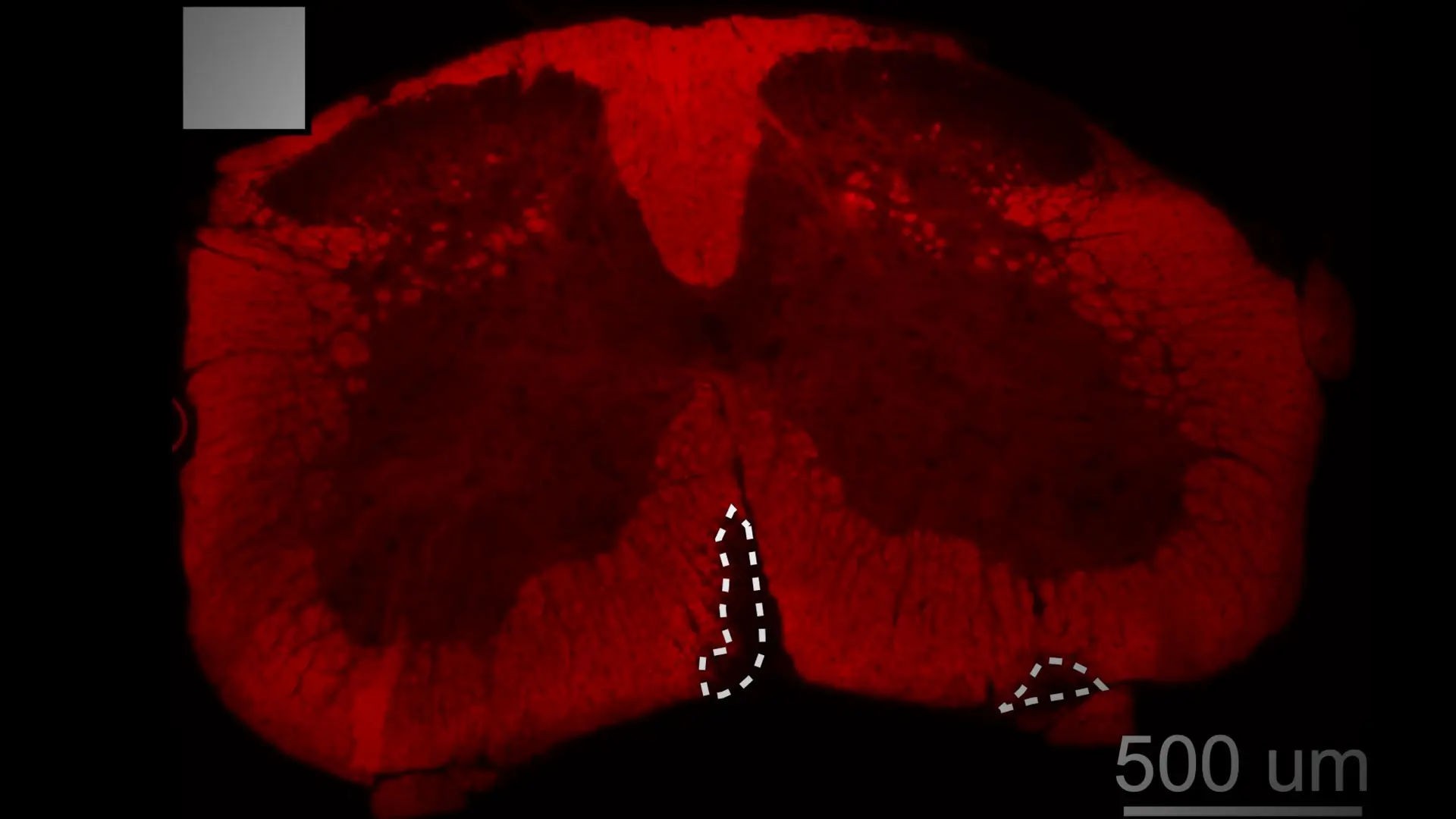
Graphical abstract of the study.

Lior Zangi, PhD, and team.
Abstract of study:
Myelopoiesis is temporally dynamic and is regulated by lifestyle to modify multiple sclerosis. Nature Communications, April 2025
Monocytes and neutrophils from the myeloid lineage contribute to multiple sclerosis (MS), but the dynamics of myelopoiesis during MS are unclear. Here we uncover a disease stage-specific relationship between lifestyle, myelopoiesis and neuroinflammation. In mice with relapsing-remitting experimental autoimmune encephalomyelitis (RR-EAE), myelopoiesis in the femur, vertebrae, and spleen is elevated prior to disease onset and during remission, preceding the peaks of clinical disability and neuroinflammation. In progressive EAE (P-EAE), vertebral myelopoiesis rises steadily throughout disease, while femur and splenic myelopoiesis is elevated early before waning later during disease height. In parallel, sleep disruption or hyperlipidemia and cardiometabolic syndrome augment M-CSF generation and multi-organ myelopoiesis to worsen P-EAE clinical symptoms, neuroinflammation, and spinal cord demyelination, with M-CSF blockade abrogating these symptoms. Lastly, results from a previous trial show that Mediterranean diet restrains myelopoietic activity and myeloid lineage progenitor skewing and improves clinical symptomology of MS. Together, our data suggest that myelopoiesis in MS is dynamic and dependent on disease stage and location, and that lifestyle factors modulate disease by influencing M-CSF-mediated myelopoiesis.
Notes From the Investigator: Cameron McAlpine, PhD
Diseases of the heart and brain are often comorbid. Indeed, individuals with multiple sclerosis, a degenerative and inflammatory disorder of the central nervous system (CNS), are at higher risk of cardiovascular complications. Moreover, people with MS and a compromised cardiovascular system experience accelerated motor function loss and more frequent episodes of MS relapse. However, how cardiometabolic syndrome, and its associated lifestyle behaviors like disrupted sleep, modify MS severity has remained largely unknown.
We used mouse models and a human trial to test if hematopoiesis, the process by which immune cells, including monocytes, are produced, links cardiometabolic syndrome and sleep with MS and neuroinflammation. We discovered that in two distinct models of murine MS, waves of hematopoiesis and monocyte generation precede and predict episodes of motor decline, monocyte accumulation in the CNS, and clinical relapse. In MS mice with cardiometabolic syndrome or disrupted sleep, myeloid hematopoiesis was heightened, and so was CNS demyelination, motor decline, and neuroinflammation. Mechanistically, we discovered that the hematopoietic growth factor M-CSF mediated these effects, and when it was blocked, monocyte production, hematopoiesis, and demyelination were reduced, and motor function was improved in mice with MS and cardiometabolic syndrome or disrupted sleep.
Finally, in a human trial, we discovered that in people with MS, consuming a Mediterranean diet over six months diminished circulating lipids, restricted myeloid hematopoiesis, and improved neurological motor function. Together, our translation data uncover new mechanistic links between cardiovascular and neurological diseases. Cardiometabolic syndrome or disrupted sleep activate myeloid hematopoiesis and the generation of monocytes, which infiltrate the CNS to perpetuate MS severity. Our findings offer the opportunity to diminish peripheral inflammation to improve the quality of life for people with MS and cardiovascular disease and to measure blood monocyte levels, which may predict episodes of MS relapse.
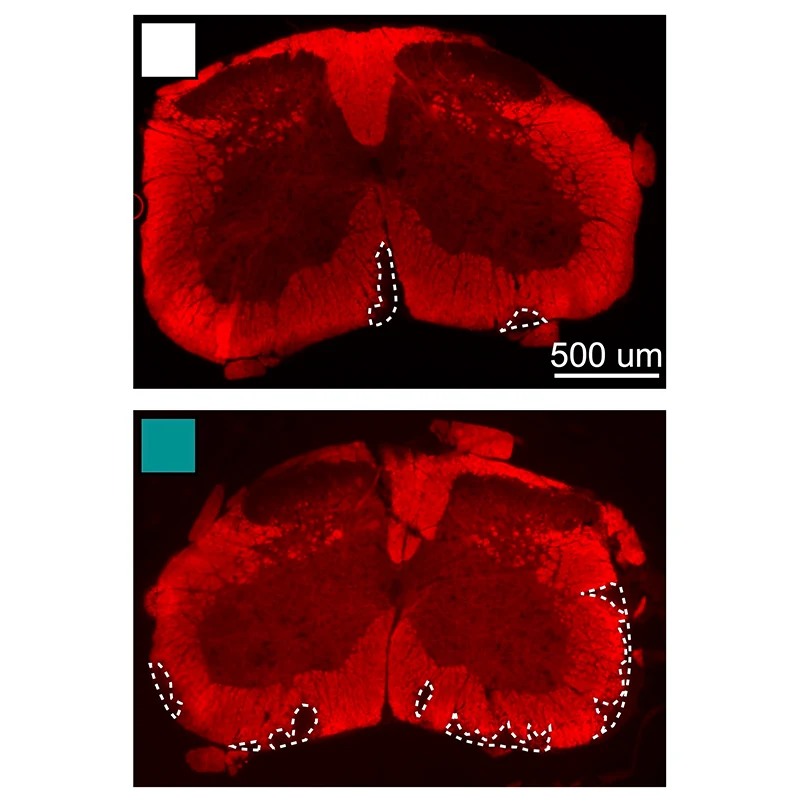
Demyelination in the spinal cord of a mouse with MS and cardiometabolic syndrome.

Cameron McAlpine, PhD, and Abi Yates, PhD, a postdoctoral fellow and first author of the study.
Abstract of Study:
Long-lasting sex-specific alteration in left ventricular cardiac transcriptome following gamma and simGCRsim radiation. Science Reports, February 2025
Space irradiation (IR) is an important health risk for deep-space missions. We reported heart failure with preserved ejection fraction like cardiac phenotype 660- days following exposure to a single dose of a simplified galactic cosmic ray simulation (simGCRsim) only in males with functional and structural impairment in left ventricular (LV) function. This sex-based dichotomy prompted us to investigate sex-specific changes in the LV transcriptome in three-month-old male and female mice exposed to Cs-γ- or simGCRsim-IR. Non-IR male and female (10 each) mice served as controls. LVs were collected at 440/660- and 440/550-days post-IR, male and female, respectively. RNA sequencing, differential gene expression, and functional annotation were performed on tissues from five mice/group. Sex and post-IR time points had the greatest influence on gene expression, surpassing the IR-type effects. SimGCRsim-IR showed more persistent transcriptome changes than γ-IR. We suggest that the single IR effects can persist up to 550-660 days, with overwhelmingly sex-biased responses at individual gene-expression level.
Notes From the Investigator: David Goukassian, MD, PhD
Deep-space missions expose astronauts to ionizing space radiation, a major health risk. The heart is a critical organ. Our previous study in the wild type mouse model showed that a single dose of a simulated galactic cosmic ray (simGCRsim) five ion (proton, oxygen, carbon, silicone, and iron) radiation caused a specific type of heart failure (heart failure with preserved ejection fraction, or HFpEF-like cardiac phenotype) only in male mice, approximately 660 days post-exposure. In the current study, we wanted to understand the biological basis for this sex-based dichotomy by analyzing how gene expression (the transcriptome) in the left ventricle (LV) of the heart changes after radiation exposure in both male and female mice. Next-generation RNA sequencing (RNA-seq) was used to study differential gene expression and functional changes.
The long-lasting changes in the transcriptome were more persistent following the simGCRsim-radiation exposure than the gamma-radiation exposure. This suggests that the high-energy particles in cosmic rays cause more sustained molecular damage or adaptation. The effects of a single dose of radiation can persist for an extremely long time—550 to 660 days after the exposure. Compared to human years, 550 to 660 days are approximately 56 to 69 plus years of age. The response at the individual gene-expression level was overwhelmingly sex-biased. This strongly supports the initial observation that males and females respond very differently to this type of space radiation, likely due to differences in gene regulation.
The findings highlight that risk assessments for deep-space missions cannot be universal; they must account for sex-specific differences in cardiac vulnerability to cosmic rays. The differential gene expression provides molecular clues (the specific genes and pathways affected) to explain why only male mice developed the HFpEF-like phenotype, paving the way for targeted countermeasures. The persistence of the transcriptional changes for nearly two years post-exposure emphasizes the need for long-term health monitoring of astronauts long after their missions are complete.
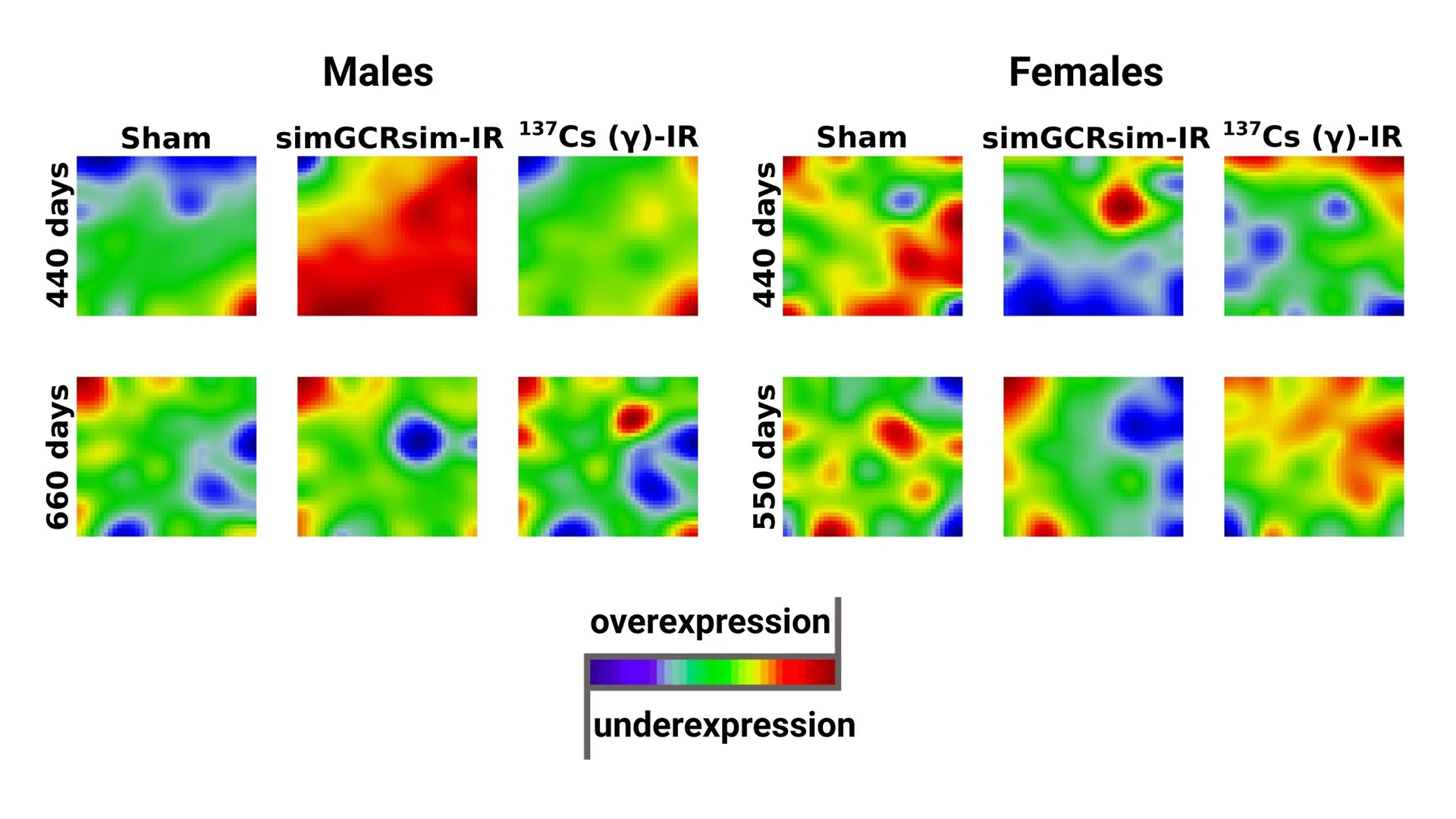
Visualization of the transcriptome landscapes studied animal groups. Inspection of portraits showed considerable differences in gene expression profiles, depending on all studied variables (sex: 5 male/ 5 female mice per group; time point: 440 days/550 days/660 days post-irradiation; irradiation: Sham/simGCRsim/γ), which was especially noticeable between males and females exposed to simGCRsim radiation at 440 days post-irradiation.

From left: Gisane Lazaryan, Siras Hakobyan, PhD, Arsen Arakelyan, PhD, and Roksana Zakharyan.