A three-drug “polypill” was found effective in preventing adverse events such as heart attacks or stroke in people age 65 and older who have previously had a heart attack, reducing cardiovascular mortality by 33 percent in this patient population. The milestone findings came from the SECURE trial led by Valentin Fuster, MD, PhD, President of Mount Sinai Heart and Physician-in-Chief of The Mount Sinai Hospital.
The study results were announced in August 2022 at the European Society of Cardiology Congress in Barcelona, Spain, and published in The New England Journal of Medicine. In December, the American Heart Association named the study one of the top advances of 2022 in cardiovascular disease research.
“The results of the SECURE study show for the first time that the polypill achieves clinically relevant reductions in the recurrent cardiovascular events among people who have recovered from a previous heart attack because of better adherence to this simplified approach with a simple polypill, rather than taking them separately as is conventional,” says Dr. Fuster, who is General Director of the Spanish National Center for Cardiovascular Research (CNIC), which developed the polypill.
For patients recovering from a heart attack, standard therapy includes three different drugs: an antiplatelet agent, such as aspirin; ramipril or a similar drug to control blood pressure; and a lipid-reducing drug, such as a statin. However, fewer than 50 percent of patients consistently adhere to their medication regimen.
“Although most patients initially adhere to treatment after an acute event such as an infarction, adherence drops off after the first few months. Our goal was to have an impact right from the start, and most of the patients in the study began taking a simple polypill in the first week after having a heart attack,” says Dr. Fuster, principal investigator of the study.
Co-author Deepak L. Bhatt, MD, MPH, who in December 2022 joined the Icahn School of Medicine at Mount Sinai as Director of Mount Sinai Heart, says, “This groundbreaking trial led by Dr. Fuster is one of the biggest advances in cardiovascular prevention of the past two decades.”
“Adherence to treatment after an acute myocardial infarction is essential for effective secondary prevention,” says José María Castellano, MD, the study’s first author and Scientific Director of Fundación de Investigación HM Hospitales.
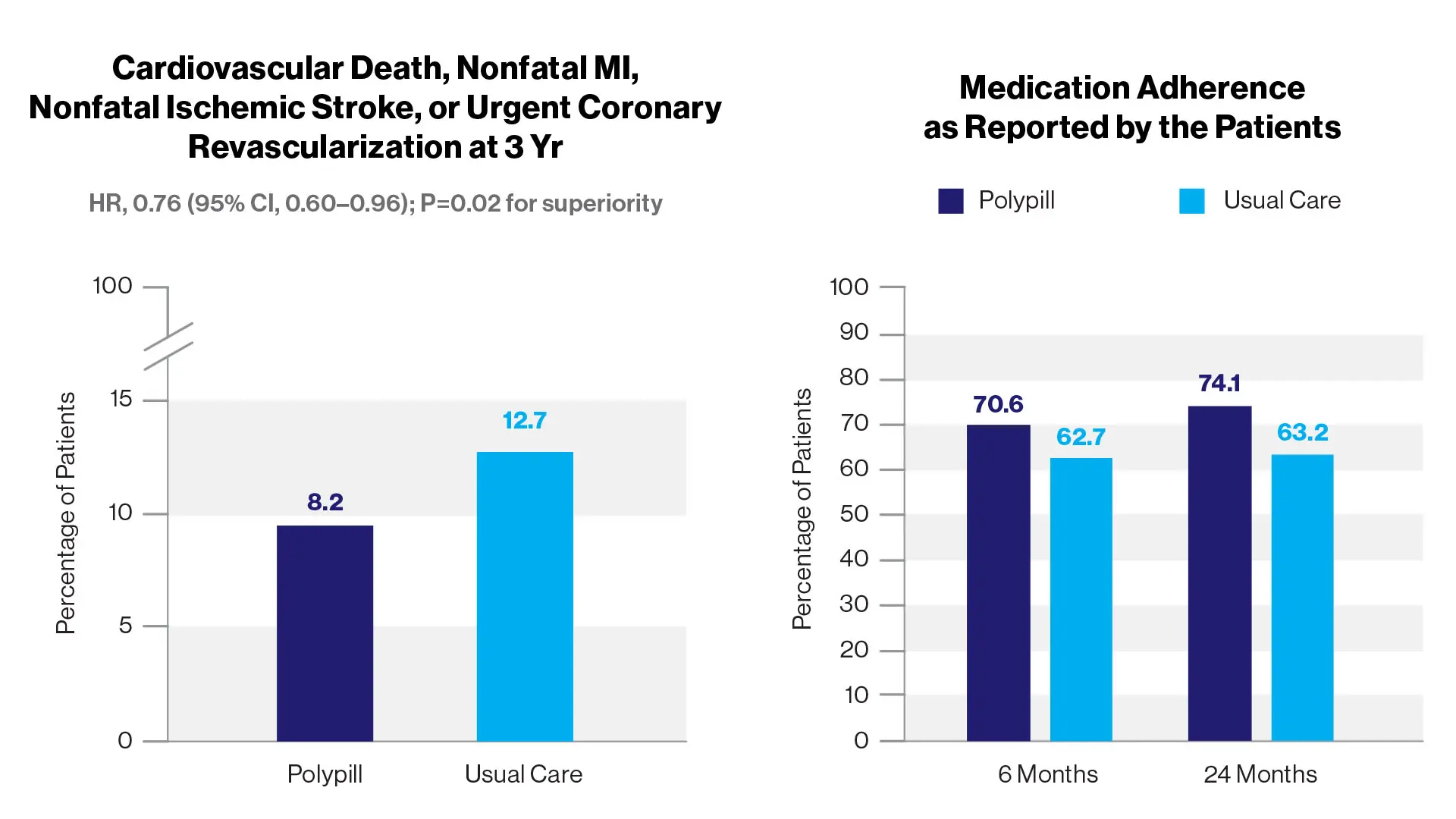
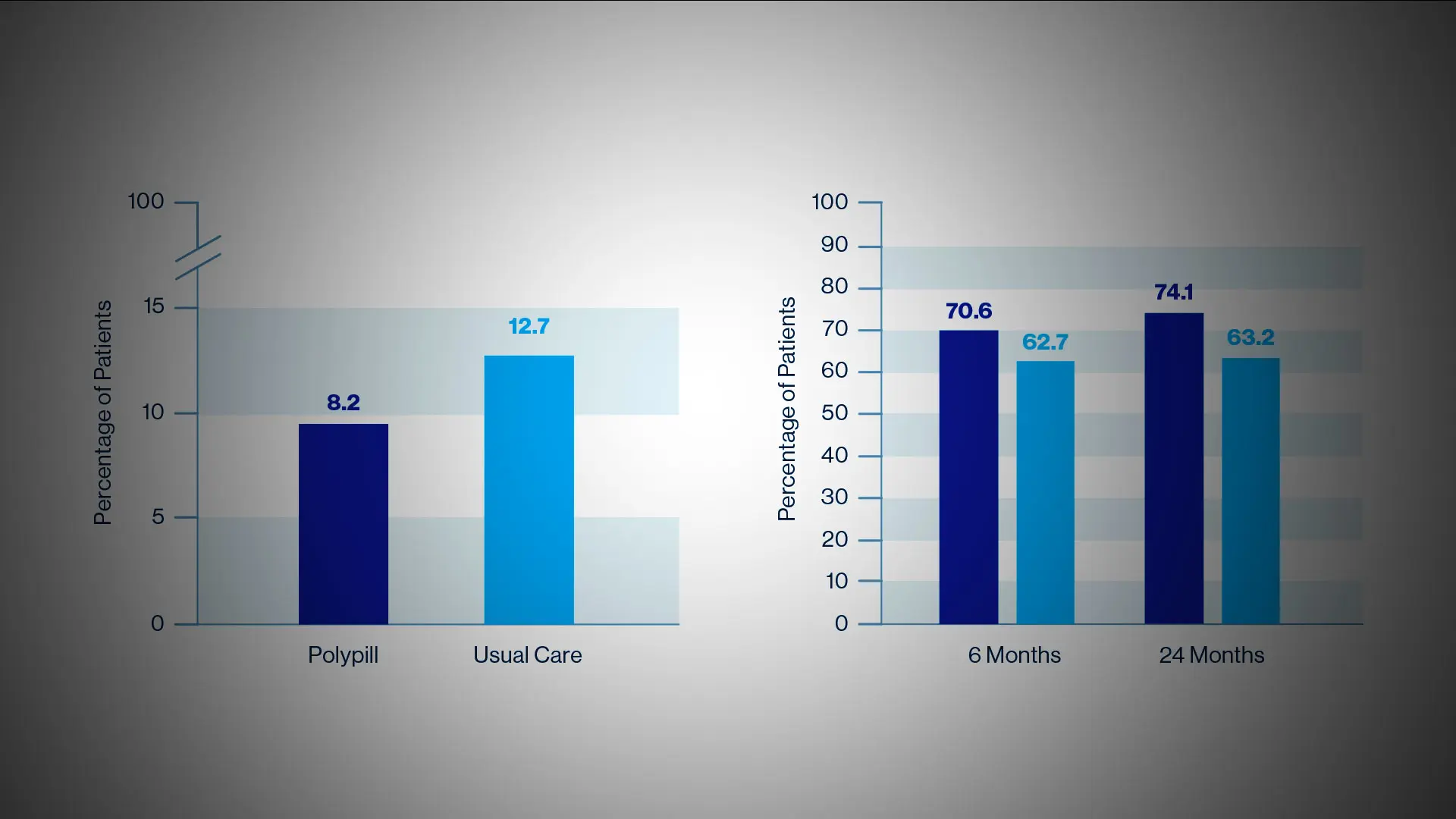
Among older adults with recent myocardial infarction, treatment with a polypill containing aspirin, ramipril, and atorvastatin resulted in a significantly lower risk of major
adverse cardiovascular events than usual care.
In the phase 3, randomized, controlled clinical trial, 2,499 patients with myocardial infarction within the previous six months were assigned to a polypill-based strategy or usual care. The polypill treatment consisted of aspirin (100 mg), ramipril (2.5, 5, or 10 mg), and atorvastatin (20 or 40 mg). The primary composite outcome was cardiovascular death, nonfatal type 1 myocardial infarction, nonfatal ischemic stroke, or urgent revascularization. The key secondary endpoint was a composite of cardiovascular death, nonfatal type 1 myocardial infarction, or nonfatal ischemic stroke.
The SECURE trial’s participants were from seven European countries (Spain, Italy, Germany, the Czech Republic, France, Poland, and Hungary). Their average age was 76 years, and 31 percent were women. The study population included 77.9 percent with hypertension, 57.4 percent with diabetes, and 51.3 percent with a history of smoking tobacco.
Researchers analyzed the incidence of four major cardiovascular events: death from cardiovascular causes, nonfatal myocardial infarction, nonfatal stroke, and need for emergency coronary revascularization. The study followed patients for an average of three years and produced conclusive results: patients taking the CNIC polypills had a 24 percent lower risk of these four events than patients taking the three separate drugs.
The standout finding of the study is the effect of the polypill on the key outcome of cardiovascular-related death, which showed a relative reduction of 33 percent, from 71 patients in the group receiving standard treatment to just 48 in the polypill group. Importantly, the study found that patients in the polypill group had a higher level of treatment adherence than those in the control group, thus confirming the findings of the earlier FOCUS study, and in part such good adherence appears to explain the benefits of the simple polypill.
The concept of a polypill for cardiovascular disease prevention was proposed in 2003 and widely debated among experts, with some arguing that it could reduce heart disease at a population level and others arguing that patients could wrongly consider it as a substitute for healthy lifestyles. In 2007, the potential value of applying the polypill strategy in high-risk patients was recognized by the WHO and the World Heart Federation, and Dr. Fuster authored a call to action in Nature Clinical Practice Cardiovascular Medicine, “A polypill for secondary prevention: time to move from intellectual debate to action.”
Scientists at the CNIC, in partnership with FERRER Laboratories, developed a polypill and have conducted a range of studies over the intervening years. CNIC scientists first demonstrated that prescription of the CNIC polypill significantly improved treatment adherence among patients recovering after a myocardial infarction, in the FOCUS study, published in the Journal of the American College of Cardiology.
The CNIC team launched the SECURE study to determine whether the improved treatment adherence with the polypill translated into a reduction in cardiovascular events. The polypill analyzed in the study was commercialized under the name Trinomia.
“The SECURE study findings suggest that the polypill could become an integral element of strategies to prevent recurrent cardiovascular events in patients who have had a heart attack,” Dr. Fuster says. “By simplifying treatment and improving adherence, this approach has the potential to reduce the risk of recurrent cardiovascular disease and death on a global scale.”
Featured

Valentin Fuster, MD, PhD
President of Mount Sinai Fuster Heart Hospital, and Physician-in-Chief of The Mount Sinai Hospital