The anticoagulant edoxaban may be just as effective as warfarin for preventing heart
attack or stroke in patients with atrial fibrillation (AF) who undergo transcatheter aortic
valve replacement (TAVR), according to a large-scale international study led by Mount
Sinai Heart
The ENVISAGE-TAVI AF trial is the largest to investigate an edoxaban-based strategy
in this patient population compared with warfarin, which is currently the standard of
care. It demonstrates that edoxaban is noninferior to warfarin. The results, which could
lead to an alternative treatment strategy, were announced in August 2021 as a late-
breaking clinical trial at the 2021 Congress of the European Society of Cardiology and
simultaneously published in The New England Journal of Medicine.
“Atrial fibrillation after transcatheter aortic valve replacement is common, especially
among older patients, but there has been little research on the optimal treatment
strategies, and this has resulted in heterogeneous use of anticoagulants in clinical
practice,” says lead investigator George Dangas, MD, PhD, Professor of Medicine
(Cardiology) and Director of Cardiovascular Innovation at the Zena and Michael A.
Wiener Cardiovascular Institute at the Icahn School of Medicine at Mount Sinai.
Mount Sinai researchers led an international trial across 173 centers in 14 countries to
compare the safety and efficacy of edoxaban versus warfarin in AF patients who need
oral anticoagulation. To determine efficacy, they looked at combined adverse clinical
events including all-cause death, thromboembolic events, and major bleeding. For
safety they looked at serious bleeding events. Investigators randomized 1,426 patients
five to 12 days after TAVR to receive either warfarin (with or without antiplatelet therapy)
or 60 mg daily oral edoxaban (with or without antiplatelet therapy).
“Atrial fibrillation after transcatheter aortic valve replacement is common, especially among older patients, but there has been little research on the optimal treatment strategies.”
- George Dangas, MD, PhD
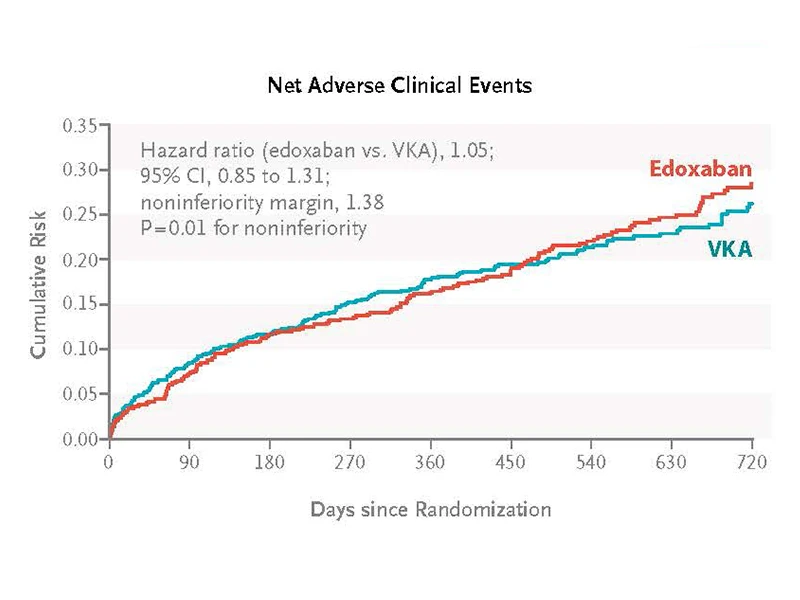
A prospective, randomized trial examined the safety and efficacy of edoxaban compared with vitamin K antagonists in patients with atrial fibrillation after TAVR.
During a median follow-up of 18 months, edoxaban was noninferior to vitamin K antagonists for preventing adverse clinical events.
They followed up with patients for up to three years after TAVR (average of 18 months).
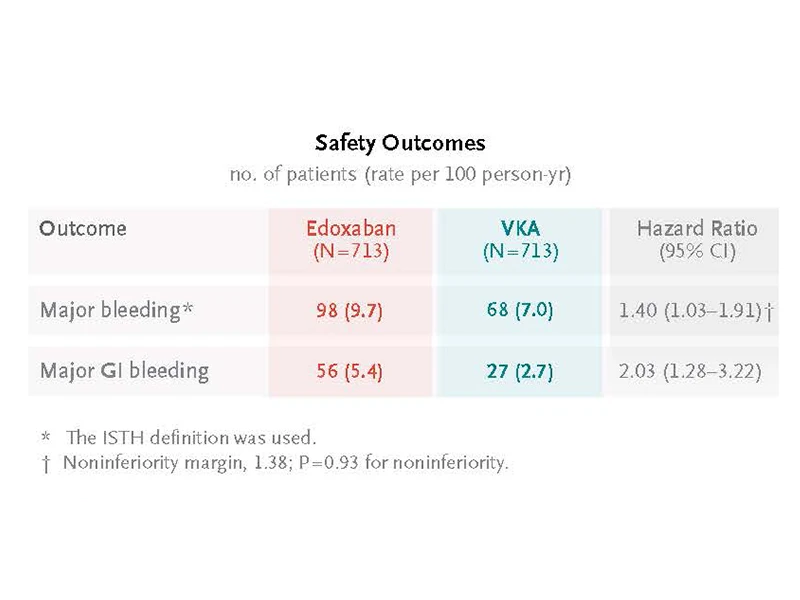
The study findings showed that edoxaban was noninferior to warfarin for efficacy as
assessed by a composite set of ischemic and bleeding adverse events. At the same
time, edoxaban led to some higher bleeding complications (mainly gastrointestinal)
when compared to warfarin (or its analogs as available in each country).
TAVR patients are typically very elderly and possess numerous comorbidities; therefore
they are at high risk for all sorts of adverse events, both ischemic and bleeding, Dr.
Dangas says. “It is important to further understand what treatment is most effective to
prevent devastating complications.” Based on the results of the trial, the primary
endpoint of non-inferiority was met, and edoxaban may be a plausible alternative to
warfarin, though with attention to increased bleeding.
Cardiologists prescribe antithrombotic agents—specifically warfarin, a vitamin K
antagonist—to prevent thromboembolic complications in patients who have TAVR, a
minimally invasive heart procedure to replace the aortic valve that is also known as
transcatheter aortic valve implantation (TAVI). Choosing the optimal antithrombotic
regimen remains challenging, especially in patients with an underlying condition like AF
that requires the use of anticoagulants. Past studies have shown that 20 to 40 percent
of TAVR patients have AF, and a large proportion of them are frail, so a main challenge
in managing their care involves balancing the risks of bleeding and stroke. Before the
ENVISAGE trial, there had been minimal research into the optimal oral anticoagulation
therapy in TAVR patients.
The next step would be to establish in large randomized trials the optimal anticoagulant
dose according to different bleeding-ischemic risk profiles, Dr. Dangas says. “It seems
that lowering the edoxaban dosage when indicated and avoiding patients with
mandatory antiplatelet therapy because of their elevated bleeding risk is reasonable
safety advice from the clinical point of view,” he says. “We will be conducting a detailed
analysis on various types of bleeding in the near future. ENVISAGE-TAVI AF suggests
that treatment with edoxaban can be valuable in the management of this high-risk
population of AF patients after TAVI.”
ENVISAGE-TAVI AF was sponsored by Daiichi Sankyo Inc. with a scientific
collaboration between scientists of Icahn Mount Sinai and Global Specialty Medical Affairs of Daiichi Sankyo.
Featured

George Dangas, MD, PhD
Professor of Medicine (Cardiology) and Director of Cardiovascular Innovation at the Zena and Michael A. Wiener Cardiovascular Institute