The Food and Drug Administration (FDA) in February 2021 approved evinacumab as an add-on treatment for patients aged 12 years and older with homozygous familial hypercholesterolemia (HoFH), a genetic condition that causes severely high cholesterol. The approval was based on research led by Robert Rosenson, MD, Professor of Medicine (Cardiology), Icahn School of Medicine at Mount Sinai.
The drug evinacumab reduced low-density lipoprotein (LDL) cholesterol by 47 percent in patients with severe hypercholesterolemia whose condition is resistant to standard treatments, a phase 3 study from Icahn Mount Sinai and other global academic sites found. Results from the study sponsored by Regeneron were published in August 2020 in The New England Journal of Medicine.
Evinacumab is a fully human monoclonal antibody that works through a different mechanism than existing drugs to bring dangerously high cholesterol to normal levels when combined with maximally tolerated lipid-lowering therapies in people with familial hypercholesterolemia, a common inherited condition that is difficult to treat.
“Our study of evinacumab shows that it can lower LDL cholesterol by half in patients unable to attain target guidelines despite maximally tolerated lipid-lowering therapy,” says Dr. Rosenson, principal investigator of the study and Director of Cardiometabolic Disorders at Icahn Mount Sinai. “Evinacumab inhibits angiopoietin-like protein 3 (ANGPLT3) and lowers LDL cholesterol through an LDL receptor independent pathway. Genetic studies have shown that people who are missing or have low levels of ANGPTL3 are known to have very low lifelong levels of LDL cholesterol and rarely suffer from atherosclerotic cardiovascular disease.”
“Its approval for use in this setting now arms cardiologists with a major new add-on therapy to bring patients with HoFH to or closer to their cholesterol-lowering goal.”
- Robert Rosenson, MD
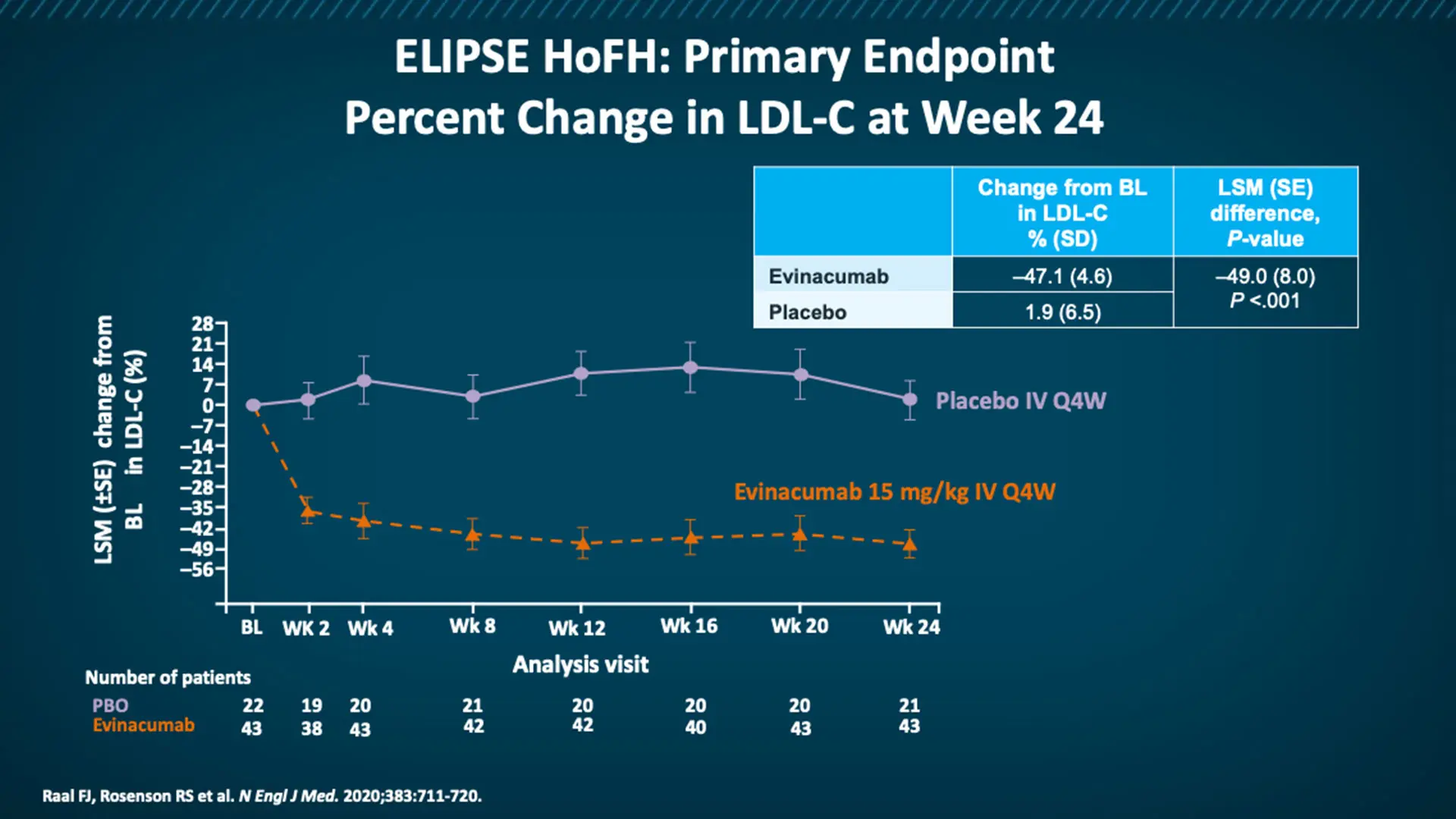
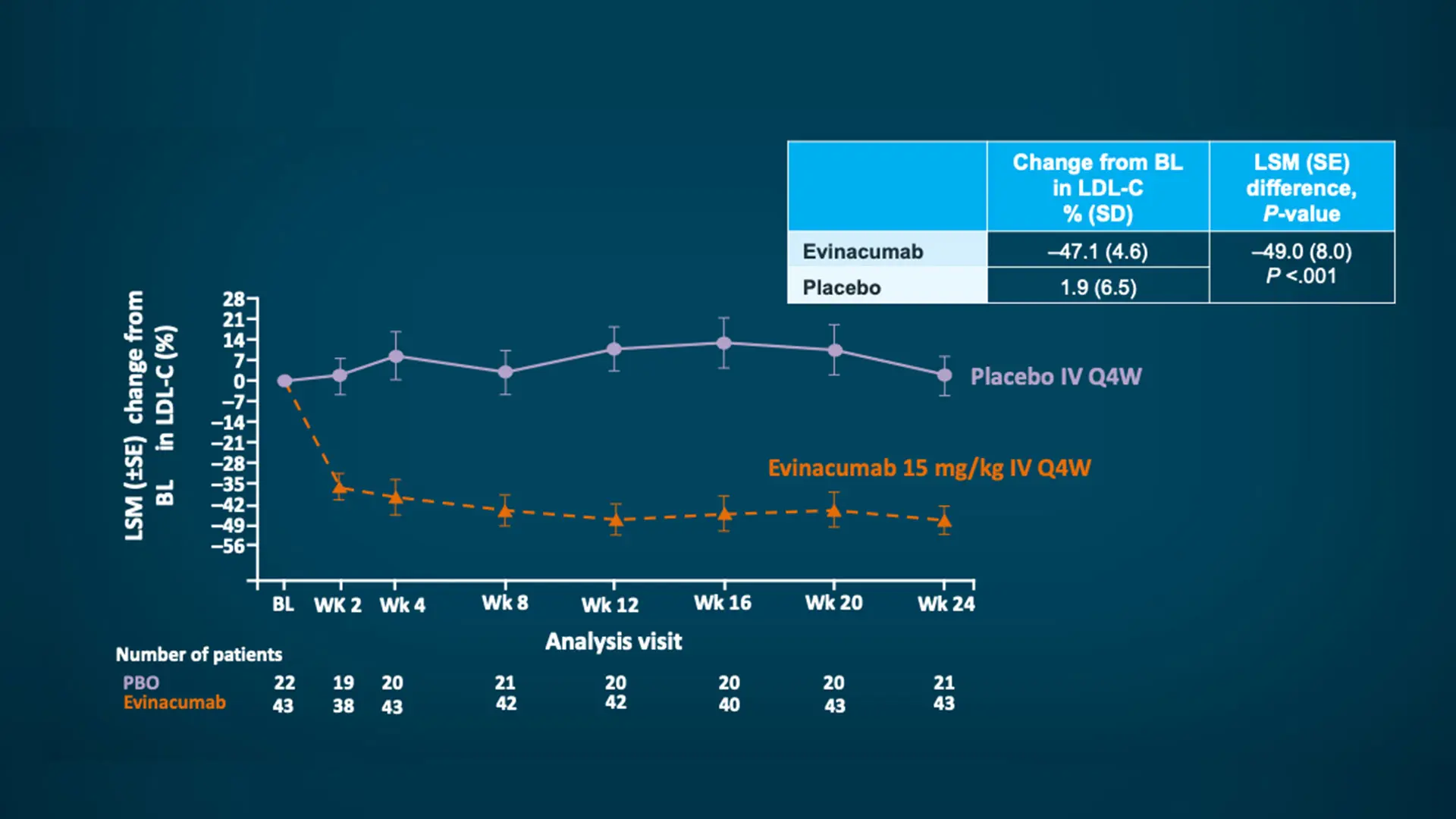
At week 24, patients in the group that received the monoclonal antibody evinacumab had a relative reduction from baseline in the LDL cholesterol level of 47.1 percent, compared with an increase of 1.9 percent in the placebo group.
Dr. Rosenson is also leading the study of evinacumab for patients with refractory hypercholesterolemia, who have high LDL cholesterol despite treatment with lipid-lowering therapies at maximum tolerated doses. Promising results of a phase 2 trial were published in December 2020 in The New England Journal of Medicine.
In the United States, approximately 7 percent of adults have been diagnosed with severe hypercholesterolemia, which is defined as having untreated LDL cholesterol at levels greater than or equal to 190 mg per deciliter. Familial hypercholesterolemia is present in 1 in 313 people, but it is far more common in patients with early-onset cardiovascular disease, occurring in 1 in 15. The 2018 American Heart Association/American Congress of Cardiology recommends an LDL cholesterol goal of less than or equal to 70 mg per deciliter in patients with very high risk of atherosclerotic cardiovascular disease, and more aggressive targets have been established by the European Society of Cardiology guideline, with recommendations to lower LDL cholesterol to 55 mg/dL or below. These goals have proven difficult for patients with hypercholesterolemia to reach through the standard “triple therapy” of a high-intensity statin, a PCSK9 inhibitor, and ezetimibe, a drug that limits the absorption of cholesterol from the intestine.
In the double-blind, placebo-controlled, phase 3 trial, researchers randomly assigned in a 2:1 ratio 65 patients with homozygous familial hypercholesterolemia who were receiving stable lipid-lowering therapy to receive an intravenous infusion of evinacumab (at a dose of 15 mg per kilogram of body weight) every four weeks or placebo. The primary outcome was the percent change from baseline in the LDL cholesterol level at week 24.
The mean baseline LDL cholesterol level in the two groups was 255.1 mg per deciliter, despite the receipt of maximum doses of background lipid-lowering therapy. At week 24, patients in the evinacumab group had a relative reduction from baseline in the LDL cholesterol level of 47.1 percent, compared with an increase of 1.9 percent in the placebo group.
“Our study demonstrated that a regimen of either subcutaneous or intravenous evinacumab can have a significant impact on LDL cholesterol,” notes Dr. Rosenson. “Its approval for use in this setting now arms cardiologists with a major new add-on therapy to bring patients with HoFH to or closer to their cholesterol-lowering goal.”
Featured

Robert Rosenson, MD
Rony Shimony Distinguished Professor of Cardiovascular Medicine, and Director of Metabolism and Lipids